Gas sensor
Introduction
The semiconductor gas sensor for carbon monoxide/combustible gas detection uses a gas-sensitive material, tin dioxide (SnO2), which has a low conductivity in clean air. The high and low-temperature cycle detection method is used to detect carbon monoxide at low temperature (1.5V heating), where the conductivity of the sensor increases with the concentration of carbon monoxide gas in the air, and to detect the combustible gases methane and propane at high temperature (5.0V heating), and to clean up the stray gases that are adsorbed at low temperature. A simple circuit is used to convert the change in conductivity to an output signal corresponding to the concentration of the gas.
Team Members
Fu Xinming, Ge Yexiao, Yang Haoqing
Setup
The diagram above shows the basic test circuit for the sensor. The sensor requires two voltages to be applied: the heater voltage (VH) and the test voltage (VC). VH is used to provide the sensor with a specific operating temperature and VC is used to determine the voltage (VRL) across the load resistor (RL) connected in series with the sensor. VC is used to determine the voltage (VRL) across the load resistor (RL) in series with the sensor. The sensor has a slight VC requires a DC power supply. VC and VH can be used as long as the electrical requirements of the sensor are met. VC and VH can share the same power supply circuit if the electrical requirements of the sensor are met. To take advantage of the sensor's performance of the sensor, it is necessary to select the appropriate RL value.
Equipment
Gas Sensitive Components
One-chip computer/microcontroller
ADC conversion module
Datasheets
Gas Sensor ENS160_Datasheet
Temperature Sensor Temperature sensor
Timeline and Milestone
| Week 8 (3/4 — 3/8) | Debug sensor module hardware devices and software code | |
| Week 9 — Week 10(3/11 — 3/22) | Measurements using gas sensors to obtain a database | |
| Week 11 — Week 12(3/25 — 4/5) | Fit curve to obtain the corresponding relationship between the output value and the gas concentration | |
| Week 13 (4/8 — 4/12) | Analyze experimental results and complete reports |
Expected Results
We hope to obtain a curve between the output value and the corresponding gas concentration, so as to know the pollution situation in the environment relatively accurately.
Report
Introduction
Band Theory
Band theory explains that within a crystal lattice, there exists a valence band and a conduction band separated by an energy gap, which is closely related to the Fermi level. The Fermi level is defined as the highest energy level occupied by electrons at absolute zero temperature. Based on the size of the energy gap between these bands, materials are classified into three main categories:
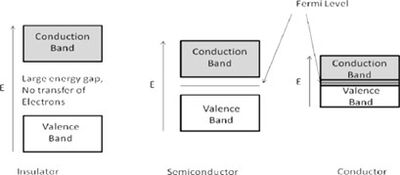
Insulators: These materials have a substantial gap between the valence and conduction bands, typically around 10 eV or more. Due to this large gap, a significant amount of energy is needed to move an electron into the conduction band, resulting in negligible electronic conduction. At zero temperature, the Fermi level represents the highest filled state.
Semiconductors: These materials feature an energy gap ranging from about 0.5 to 5.0 eV. Below the Fermi level, electrons do not have enough energy to bridge this gap, preventing conduction. However, once the energy exceeds the Fermi level, electrons can transition to the conduction band, leading to increased electrical conductivity.
Conductors: In conductors, the Fermi level is positioned within the conduction band itself, allowing for continuous electron flow and high conductivity.
Band Theory in Gas Sensors
Band theory has played a crucial role in understanding the functionality of gas sensors for several years. In this context, the interaction between a target gas and a metal oxide film's surface—typically involving surface adsorbed oxygen ions—causes a shift in the material's charge carrier concentration. This shift subsequently affects the material's conductivity or resistivity.
For n-type materials, oxidizing gases increase the resistance of the thin film, while reducing gases decrease it. The opposite is true for p-type materials. This interaction, involving oxidation-reduction (REDOX) reactions, alongside the transduction function, defines the chemiresistive behavior of these sensors. Metal oxide sensors are commonly configured as sintered pellets or as thick and thin films. The specific geometry of the crystallites and their interconnections significantly influence these receptor and transduction functions, highlighting the impact of nanotechnology in enhancing sensor performance by manipulating materials at the atomic and molecular scale.
On the other hand, p-type semiconductors, where positive holes are the majority charge carriers, exhibit increased conductivity in the presence of an oxidizing gas, which augments the number of positive holes. Conversely, the presence of a reducing gas increases the material's resistance by introducing negative charge that diminishes the concentration of positive charge carriers (holes). The various responses of these materials to different gases are summarized in Table 1.
| Classification | Oxidising Gases | Reducing Gases |
|---|---|---|
| n-type | Resistance increase | Resistance decrease |
| p-type | Resistance decrease | Resistance increase |
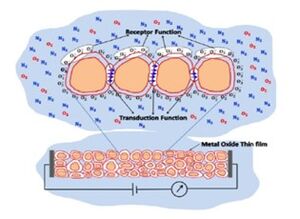
The chemiresistance of metal oxides is also linked to the width of the space charge region that forms on the crystallites due to electron transfer during the adsorption and desorption of gas molecules. This space charge region creates a potential barrier that affects conduction between the grains, leading to changes in the Fermi level. In n-type materials, the interaction with oxidizing gases causes the energy bands to bend upward as electrons are depleted, whereas with reducing gases, the bands bend downward due to electron accumulation. Thus, the space charge region and band bending are closely related to the carrier concentrations within the grain, which are influenced by the grain's size and shape.
Why carbon dioxide?
As of March 2024, carbon dioxide concentrations in the Earth's atmosphere were recorded at 425 ppm. Carbon dioxide is utilized in a variety of ways including in carbonated beverages, pneumatic systems, fire extinguishers, and as a critical component for photosynthesis. Additionally, it is used in lasers and refrigeration systems, both naturally and in human applications.
Carbon dioxide also acts as a greenhouse gas, absorbing infrared energy from the atmosphere. This absorption causes the gas to vibrate, subsequently transferring heat to the surrounding air and raising the ambient temperature. Increased levels of carbon dioxide can lead to higher temperatures. Therefore, accurate and reliable sensors that can operate within appropriate temperature ranges are crucial for monitoring changes in atmospheric carbon dioxide concentrations. The recent rise in carbon dioxide levels, primarily due to human activities such as the combustion of hydrocarbons and other carbon-containing fuels like coal, methane, petrol, and kerosene, has heightened concerns about its contribution to global warming. Such activities, when combined with oxygen, produce carbon dioxide and water. Monitoring the emissions of CO2 during these combustion processes is vital as it provides essential real-time data on the quantity of carbon dioxide being emitted.
| CO2 Concentration and Exposure time | Effect on Health (symptoms) |
|---|---|
| 0.035% | Approximate atmospheric concentration, no noticeable effect. |
| 3.3–5.4% for 15 mins | Increased depth of breathing |
| 7.5% for 15 mins | Feeling of an inability to breathe, increased pulse rate, headache, dizziness, sweating, restlessness, disorientation, and visual distortion. |
| 3%, for over 15 hours | Decreased night vision, colour sensitivity |
| 10%, 1.5 mins | Eye flickering, increased muscle activity, twitching |
| 10+% | Difficulty in breathing, impaired hearing, nausea, vomiting, a strangling sensation, sweating, after 15 minutes a loss of consciousness. |
| 30% | Unconsciousness, convulsions. Several deaths attributed to CO2 at concentrations of more than 20% |
Since 1958, atmospheric carbon dioxide levels have consistently increased and are expected to continue rising as long as there is a demand for fossil fuel combustion. High concentrations of carbon dioxide can have significant adverse health effects, including drowsiness and, at very high levels, suffocation.
The National Environmental Agency (NEA) recommends indoor CO2 levels that should ideally not exceed 800 ppm. Thus, the ability to measure and control carbon dioxide levels is crucial for maintaining indoor air quality and protecting human health.
Experimental Procedure
Temperature Sensor test
Background
In this experiment, it is essential to ensure that the TVOC and CO2 gas sensors operate accurately under various environmental temperature and humidity conditions. Since the performance of these sensors—such as range, sensitivity, and accuracy—can be significantly affected by ambient temperature and humidity, the introduction of a temperature sensor for simultaneous measurement becomes crucial. This setup allows for real-time monitoring of the experimental environment’s temperature, thus enabling the calibration of the gas sensors' readings for more accurate data.
Principle of the Temperature Sensor
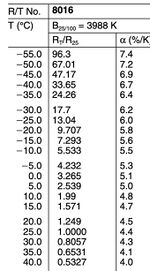
We chose the NTC thermistor, whose model No. is B57861S0103F040, as our temperature sensor. The resistance of an NTC thermistor decreases as the temperature increases. This characteristic is due to the properties of its semiconductor material, where more charge carriers (electrons and holes) participate in conduction at higher temperatures, reducing resistance. Additionally, the NTC thermistor used in the experiment has a specific B value, an important parameter describing its temperature sensitivity. The B value, determined between two specific temperatures (usually 25°C and 100°C), characterizes the rate at which the resistance changes with temperature. With a B value of 3988K and a nominal resistance of 10kΩ at 25°C, this sensor demonstrates sensitivity to changes within a specific temperature range.
Test Process
Prior to the experiments, under the help of professor, we attached the NTC thermistor to a cable using soldering techniques, being mindful to control the soldering temperature and duration to prevent overheating damage. After soldering, we measured the sensor’s resistance with a multimeter and converted these readings to temperatures using the resistance-temperature (R/T) characteristic curve from the datasheet as shown
Experimental Verification

As illustrated in the experimental design diagram, we tested the temperature sensor in a beaker containing dry ice. The sublimation of dry ice absorbs heat from its surroundings, reducing the temperature inside the beaker, thereby providing a dynamic environment to test the sensor's responsiveness and accuracy. Preliminary measurements indicated a significant drop in ambient temperature, approximately 5°C, demonstrating the sensor's capability to accurately respond to low-temperature changes.
Gas Sensor Calibration
After verifying the performance and reliability of the temperature sensor, we proceeded to calibrate another key component in the experiment—the gas sensors. The primary goal here was to ensure proper interfacing and communication between the gas sensors and a microcontroller, thus providing a reliable hardware foundation for accurate CO2 concentration measurement.
Hardware Connections

We followed the design schematics to make connections between the gas sensors and an Arduino microcontroller. This included correctly attaching the sensor’s data output pins to the appropriate input ports on the microcontroller and ensuring proper power and ground connections. The accuracy of these connections is crucial for the success of the experiment, so we carefully checked all connection points to ensure they were correct. The final connection situation is shown in Figure 3.2。
Software Configuration
Once the hardware connections were completed, we connected the Arduino microcontroller to a computer via a USB interface. On the computer, we launched the Arduino IDE software and loaded code specifically designed for testing CO2 concentrations. This software environment allowed us to upload code to the microcontroller and monitor the running program and data output from the sensors.
Communication Parameter Adjustment
After initially running the test code, we discovered that minor adjustments to the communication addresses were necessary to ensure stable and reliable data transfer between the microcontroller and the gas sensors. After adjusting these parameters, we approached the sensor and exhaled near it to test its responsiveness to changes in CO2 concentration. This helped us evaluate the sensor's reaction speed and accuracy to environmental changes.
System Testing and Setup
After confirming that the hardware connections were correct and the software could control the sensor accurately, we conducted a series of functional tests on the sensors. For example, we hold the sensor and exhale it from far to near. The results indicated that all hardware and software were functioning properly, and the sensors' sensitivity and response characteristics met the experimental requirements. Subsequently, we began constructing the final experimental setup, preparing to enter the data collection phase.
Data Collection
Experimental Purpose
The aim of this experiment is to assess the performance of the gas sensors under different CO2 concentration environments. By precisely controlling the experimental conditions, we can determine the specific impact of CO2 concentration on the output of the gas sensors, as well as evaluate the effect of temperature changes on the rate of CO2 gasification.
Experimental Materials and Setup

Materials needed for the experiment include dry ice, a beaker, tape, the temperature sensor, cable, multimeter, and gas sensors. Initially, we placed the dry ice in the beaker and sealed the beaker's mouth with tape to control the CO2 concentration inside. The temperature and gas sensors were placed together inside the beaker to simultaneously monitor temperature and CO2 levels.
Preliminary Observations and Adjustments
In the early stages of the experiment, the gas sensor recorded CO2 concentrations close to its lower measurement threshold, fluctuating slightly. Data from the temperature sensor indicated that the temperature inside the beaker was low. This observation led us to conclude that the rate of dry ice sublimation was significantly influenced by temperature, necessitating an increase in the beaker's temperature to accelerate the transformation of dry ice into CO2 gas.
Optimization of Experimental Process
To enhance CO2 output and test the gas sensor’s performance at different concentrations, we heated the beaker. As the temperature increased, the rate of dry ice sublimation accelerated, significantly raising the CO2 concentration inside the beaker. At this point, the gas sensor began to record higher CO2 concentrations, with increased data range and variability.
Data Measurement and Collection
After heating, we continued to collect data from the gas and temperature sensors to monitor changes in CO2 concentration under heated conditions and its impact on sensor performance. The collected data included real-time temperature changes, variations in CO2 concentration, and the sensors' response times. This data will be used to analyze the sensitivity and accuracy of the gas sensors under various environmental conditions.
Preliminary results indicate that the gas sensors exhibit good responsiveness to varying CO2 concentrations, validating their potential application in environmental monitoring and control systems. Further data analysis will help us understand the behavior and stability of the sensors under extreme conditions.
Results
To verify the sensor's performance under normal room temperature atmospheric conditions, it is an important step to first measure the level of harmful organic gases in the room. These organic gases may include formaldehyde, benzene and other toxic volatile organic compounds (TVOCs). By measuring the concentrations of these gases, we can get a basic idea of the state of indoor air quality.
Then, converting the concentrations of these organic gases to equivalent carbon dioxide levels is another key measurement. The equivalent carbon dioxide level is often used to assess the ventilation of indoor air, as carbon dioxide levels are often closely related to air quality. This conversion process involves the use of specific models or formulas to estimate the equivalent carbon dioxide level based on the concentrations of organic gases.
Due to the limitations of the experimental conditions (the release of TVOCs indoors is hazardous), we preferred that varying the concentration of carbon dioxide in the air was a safe and practical way to verify the usability and sensitivity of the ENS160 gas sensor. enS160 assesses the air quality by detecting the concentration of a chemical, so controlling and varying the level of carbon dioxide can be an effective way to test the sensor's responsiveness and accuracy.
Measurement of atmospheric carbon dioxide concentration at room temperature
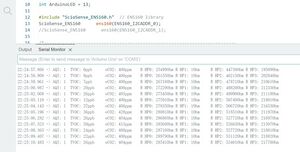
Before changing the CO2 concentration, CO2 levels were first measured under conventional room air conditions to obtain baseline data.
By measuring the baseline co2 content, we know that the air in the lab contains 400-420 ppm of co2. Typically, the atmospheric carbon dioxide content is 0.04%, or 400 ppm, but because the lab environment is relatively airtight and there are people breathing in the lab, the value is slightly higher than 400 ppm.
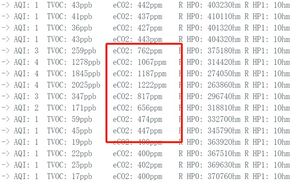
After verifying the baseline of co2, we want to know how this gas sensor works in real life applications, starting with the value of human exhaled co2 in the atmosphere.
From the data, we can see that in a closed environment, carbon dioxide from exhaling can increase the carbon dioxide concentration of the air around the sensor by a factor of 2 to 1,222 ppm, which is 0.12% carbon dioxide in the air. We know that human exhaled gas is 80% nitrogen, 13% to 16% oxygen and 4% carbon dioxide. The reason for the 33-fold difference from the measured value is analyzed for the following two reasons:
- The exhaled air is diluted in the atmosphere, even when exhaling into the sensor, because the amount of exhaled air is much smaller than the volume of indoor air, so the effect is minimal.
- Because the measurement process of co2 from tvoc through the algorithm to convert the equivalent carbon dioxide content, but the human exhaled gas, almost no toxic organic volatiles, containing carbon elements are part of the co2, may be ens160 gas sensors are not sensitive to inorganic substances containing carbon elements, this part of the test we will verify through the next.