EA Spectroscopy as a series of sensors: Investigating the Impact of Solvent Type on Mobility in Organic Diodes: Difference between revisions
| (108 intermediate revisions by the same user not shown) | |||
| Line 4: | Line 4: | ||
Liu Chenyang A0328377R | Liu Chenyang A0328377R | ||
== | ==1. Introduction== | ||
===1.1 Fundamentals of polymer organic semiconductors=== | |||
Organic semiconductors are a class of materials composed of organic molecules or polymers that possess semiconductor-like energy levels and are capable of transporting electrical charges. Compared with inorganic semiconductors, organic semiconductors offer advantages such as structural tunability, solution processability, and compatibility with flexible electronic devices[8]. Among them, polymer are particularly important because their long-chain molecular structures enable good film-forming ability and make them highly suitable for large-area solution processing. | |||
PBDB-T is a representative polymer that has attracted significant attention due to its excellent optoelectronic properties and widespread use in organic electronic devices, especially organic solar cells. Structurally, PBDB-T consists of repeating molecular units that form a conjugated polymer backbone. The conjugated backbone plays a central role in determining the semiconducting behavior of the material, as it governs the energy levels and facilitates charge transport along the polymer chain. In addition, PBDB-T contains alkyl side chains attached to the backbone. These side chains mainly improve the solubility and processability of the polymer, which are essential for solution-based fabrication methods. Therefore, the molecular structure of PBDB-T, including its repeating units, conjugated backbone, and side chains, provides the basis for understanding its aggregation behavior, film formation, and charge-transport properties. | |||
<div style="text-align: center;"> <math> I = I_0 (1 - R)^2 e^{-\alpha d} </math>----------( | [[image:Fig1_1.png|200px|center]] | ||
<center> Fig 1.1. Chemical structure of PBDB-T </center> | |||
===1.2 Basic device for organic semiconductor research=== | |||
In studies of organic semiconductors, organic diodes are fundamental devices for investigating charge-transport behavior. Typically, an organic diode consists of an ITO anode, a metal cathode, and an organic semiconductor thin film sandwiched between the two electrodes. | |||
[[image:Fig1_2.png|300px|center]] | |||
<center> Fig 1.2. Basic structure of organic diode </center> | |||
[[image:Fig1_3.png|800px|center]] | |||
<center> Fig 1.3. Energy level diagram of a hole-dominated diode at zero applied bias (left), at applied bias equals to built-in potential (<math>V_{bi}</math>) (centre) and at applied bias larger than <math>V_{bi}</math> (right). </center> | |||
To achieve the highest efficiency, we usually utilize hole injection layer and electron injection layer to make sure of the efficient charge injection at both hole and electron contacts, which is guaranteed by the match of the energy levels of these various layers. | |||
The work function is the minimum energy required to move an electron from the Fermi level to the vacuum level. The injection barrier is the energy difference that carriers must overcome in order to be injected from an electrode into the organic semiconductor. Meanwhile, in organic semiconductor diodes, an Ohmic contact refers to a contact at which the carrier injection and collection resistance at the metal/organic-semiconductor interface is lower than the bulk transport resistance of the organic semiconductor, so that device operation is no longer contact-limited but primarily bulk-transport-limited. In an organic semiconductor device, a built-in potential, Vbi, is formed even at zero applied bias because of the difference in the work functions of the two electrodes and the interfacial energy-level alignment. This built-in potential plays a central role in determining the internal electric field distribution and carrier injection behavior, and is therefore an important parameter for understanding charge transport in organic diodes. | |||
More importantly, the energy-level diagram shows the energy-level matching in the diode. It refers to the alignment between work function of the electrodes and the frontier energy levels of the organic semiconductor, as well as the resulting interfacial energy-level alignment. Such alignment determines the carrier injection and extraction barriers at the contacts. It effects the interfacial resistance and indicates whether the device is contact-limited or forms the Ohmic-contact. In general, better energy-level matching leads to smaller carrier-injection barriers and lower interfacial resistance. It also promotes Ohmic contact formation. In addition, the energy-level matching determines the built-in potential, and thus influences the internal electric field, charge transport, and overall device performance. | |||
At the same time, the energy-level diagram also reveals the operating principle of the organic diode. At zero bias, the Fermi levels of the anode and cathode are aligned. Because the two electrodes have different work functions, a built-in potential is formed, and creates an internal electric field directed from the cathode to the anode. Meanwhile, hole and electron injection are limited by their injection barriers, so carrier injection is weak and the current remains low. As the applied bias increases and compensates the built-in potential, the device reaches the flat-band condition. In this state, the energy-level bending in the organic layer disappears, and the net internal electric field becomes zero. Therefore, this condition is commonly used to characterize the built-in potential, Vbi. When the applied bias further exceeds Vbi, the device enters a clear forward-conduction regime. The energy levels then tilt further. The forward bias changes internal electric field, making it easier for carriers to be injected into the HOMO or LUMO and thereby promoting charge transport through the device. Under this condition, if the electron (hole) injection barrier is sufficiently large while the hole (electron) injection barrier is small, electron (hole) injection is effectively blocked, while holes (electrons) can be injected efficiently, resulting in unipolar diode. | |||
During the experiment, carrier transport is investigated by measuring the current density-voltage (J-V) characteristics of the diode. The J-V curve is a basic but powerful tool. It provides direct information about the charge-transport behavior in the organic semiconductor layer. At low bias, the device usually operates in the Ohmic regime, where the current increases approximately linearly with voltage. As the applied voltage increases, however, more carriers are injected into the organic layer and begin to accumulate. This accumulation forms space charge. The space charge distorts the internal electric field and limits the current, causing the device to enter the space-charge-limited current (SCLC) regime. In this regime, the current is mainly controlled by bulk transport in the organic semiconductor. Charge transport may also be affected by traps. A trap is a localized energy level within the bandgap that captures and confines charge carriers, thereby reducing their effective transport. One important feature in the J-V curve is the knee point. This point marks the transition from the Ohmic regime to the SCLC regime as the voltage increases. Therefore, by analyzing the shape of the J-V curve and the position of the knee point, it is possible to evaluate carrier transport and to identify the influence of space charge and trap effects in the device. | |||
[[image:Fig1_4.png|300px|center]] | |||
<center> Fig 1.4. Current density–voltage (J–V) curves of PM6 in CB at 60 °C, where different colors represent different pixels of the same device; the kink in the curves indicates the transition from ohmic contact to space-charge-limited current (SCLC) conduction. </center> | |||
In this study, carrier transport behavior is quantitatively evaluated in terms of carrier mobility. Mobility describes how well charge carriers move under an electric field, and it is one of the most important parameters for characterizing charge transport in organic semiconductors. A higher mobility generally indicates more efficient carrier transport within the material. Carrier mobility is influenced by multiple factors. These include backbone planarity, aggregation, side-chain structure, and other molecular or microstructural features of the polymer semiconductor. Among these factors, aggregation is particularly important because it is closely related to molecular packing and charge-transport pathways. | |||
Aggregation refers to the tendency of polymer chains, either in solution or in a film, to associate with one another rather than remain fully separated. As a result, small clusters or more ordered structures can be formed[4]. The aggregation state in solution strongly affects the film-formation process and can further influence the final charge-transport properties, including mobility. Aggregation itself can be affected by factors such as solution processing and chemical substitution. Therefore, this study mainly focuses on how aggregation influences carrier mobility, with particular attention paid to the effects of solvent processing and substitution on the aggregation behavior of the polymer system. | |||
Since aggregation plays an important role in determining carrier mobility, it is necessary to further understand the factors that govern aggregation behavior in polymer semiconductors. In this work, two key factors are considered: solvent processing and halogen substitution. These two factors influence aggregation from different perspectives. Solvent processing mainly affects the solution-state behavior of polymer chains, whereas halogen substitution changes the intrinsic molecular interactions of the polymer backbone. In the following sections, these two effects will be discussed in detail. | |||
===1.3 Solvent=== | |||
For polymer systems, solvents are generally classified as either good solvents or poor solvents. In a good solvent, strong polymer-solvent interactions enable effective solvation of the polymer chains, such that the chains adopt relatively extended conformations and remain well dispersed, with only a weak tendency toward aggregation. By contrast, in a poor solvent, polymer-solvent interactions are comparatively weak, while interchain interactions become more significant. Under these conditions, the polymer chains tend to contract, entangle, and aggregate, and may eventually precipitate from solution. | |||
For conjugated polymers, however, this binary classification is often insufficient to describe their solution behavior in a comprehensive manner. Zhao et al. introduced the concept of a borderline solvent, defined as a solvent with a solvating ability intermediate between that of a good solvent and a poor solvent. In such a medium, the polymer chains are not fully extended and uniformly dispersed, as would be expected in a good solvent. Instead, they are more prone to intermolecular approach and the formation of a certain degree of ordered aggregation, while remaining dispersed in solution without obvious precipitation. Borderline solvents therefore have particular significance in the regulation of aggregation in conjugated polymer systems, and consequently exert an important influence on subsequent film formation, thin-film morphology, and device performance. | |||
As shown in Figure 1.5, Zhao et al. proposed a mechanism by which a borderline solvent can induce ordered aggregation in conjugated polymers. In a borderline solvent, the polymer chains are more likely to undergo an early transition from coil-like conformations to more extended chain conformations, and subsequently form small, ordered 𝜋-stacking aggregates. These initial aggregates can then further organize and grow into more distinct plate-like stacked structures with a high degree of order and a preferred orientation. During the subsequent spin-coating process, the aggregates continue to spread, develop, and align on the substrate, thereby promoting the formation of an ordered thin film. Zhao et al. suggested that such dilute-solution 𝜋-stacking and the resulting extended-chain aggregates are crucial for achieving pronounced lamellar order in conjugated polymer films. | |||
By contrast, in a good solvent, polymer–solvent interactions are sufficiently strong to maintain the polymer chains in a more fully solvated and dispersed state. Under these conditions, significant 𝜋-stacking tends to occur only at a later stage of film formation. At that point, however, the polymer chains have already become more entangled, and the adjacent 𝜋-stacked aggregates are more likely to interpenetrate and entangle with one another. This suppresses the formation of extended-chain 𝜋-stacks and reduces the degree of structural order in the resulting film. Consequently, films processed from a good solvent typically contain a larger fraction of disordered or amorphous regions. | |||
These observations demonstrate that the solvent plays a decisive role in regulating polymer aggregation behavior. It influences not only the conformation and dispersion state of the polymer chains in solution, but also the onset, extent, and degree of order of aggregation during film formation. Therefore, solvent selection is a key factor in controlling polymer aggregation, and it can subsequently affect thin-film morphology and charge-transport properties. | |||
[[image:Fig4.png|400px|center]] | |||
<center> Fig 1.5. (a) Schematic of the dilute π-stacking mechanism that leads to formation of unentangled π-stacked extended-chain lamellae that give rise to the monolayer-terraced film morphology. This mechanism can be induced by a borderline solvent or in very dilute solutions of a good solvent. (b) Schematic illustration of the morphology of a thick film deposited from a good solvent. </center> | |||
===1.4 Material Choice=== | |||
Solvent processing and substitution are two key factors governing the aggregation behavior of polymers, and are therefore closely related to their charge-transport properties. Accordingly, in order to systematically investigate the influence of aggregation on mobility, it is necessary to select a representative polymer system that can both reflect substitution effects and exhibit high sensitivity to processing conditions. On this basis, the PBDB-T family polymers were chosen as the model materials in this study, including PBDB-T, PM6 (PBDB-T-2F), and PM7 (PBDB-T-2Cl). | |||
PM6 and PM7 are obtained from PBDB-T through halogen substitution, in which the two hydrogen atoms are substituted with fluorine and chlorine, respectively. This group of polymers, which share closely related molecular structures but differ in their substituents, provides an ideal model system for investigating the effects of halogen substitution on polymer aggregation and charge transport. In addition, these polymers are highly sensitive to solution-processing conditions. Their aggregation behavior, film-formation process, and resulting thin-film morphology can be readily modulated by solvent selection and processing treatment, making them particularly suitable for studying the relationship between solvent processing and aggregation. Finally, the PBDB-T family polymers are among the most representative donor polymers in the field of organic solar cells, and thus possess broad application relevance and substantial research value. Therefore, the use of the PBDB-T family as a model system not only helps elucidate the intrinsic relationship among substitution, solvent processing, aggregation, and mobility, but also ensures that the conclusions obtained are of greater general relevance and practical significance. | |||
[[image:Fig1_6.png|700px|center]] | |||
<center> Fig 1.6. Chemical structure of PBDB-T family polymers. </center> | |||
==2. Project Aim== | |||
The aim of this study is to investigate how solvent type influence the aggregation behavior and charge-transport properties of PBDB-T family polymers, including PBDB-T, PM6, and PM7. In this work, the processing conditions include solvent type, solution concentration and thermal treatment. Specifically, chlorobenzene (CB) and chloroform (CF) are used as the processing solvents. Room temperature refers to 23°C, while the heating treatment in this study is applied only to the polymer solutions, with the solution temperature set at 100°C for CB and 60°C for CF. This work investigates how these factors effect solution-state aggregation, and further, by fabricating thin films from solutions with different aggregation states, to explore how these factors influence charge transport. Meanwhile, both the solution processing and film fabrication were carried out inside a glovebox to avoid interference from air exposure. To achieve this, hole-only diodes (HODs) based on these polymers are fabricated and analyzed through their current density-voltage (J-V) characteristics. Through this study, we expect to dig deeper regarding how molecular packing, aggregation, and film morphology collectively affect carrier mobility in conjugated polymer systems. | |||
==3. Experiment methods== | |||
===3.1 Measurement of aggregation in different solvent systems=== | |||
====3.1.1 UV-Vis analysis of solution-state aggregation==== | |||
UV-Vis absorption spectroscopy is a simple and effective technique for testing aggregation behavior in polymer solutions[41][42]. The principle of UV-Vis is Beer-Lambert Law[43]: by comparing the incident light intensity I0 with the transmitted light intensity I, the transmittance T of the sample can be obtained as | |||
<div style="text-align: center;"> <math> T = \frac{I}{I_0} </math>----------(3.1)</div> | |||
The absorbance A is then given by | |||
<div style="text-align: center;"> <math> A = -\log_{10}(T) = \log_{10}\left(\frac{I_0}{I}\right) </math>----------(3.2)</div> | |||
According to the Beer-Lambert law, the absorbance of a solution is proportional to the concentration 𝑐, the optical path length 𝑙, and the molar extinction coefficient | |||
<div style="text-align: center;"> <math> A = \varepsilon lc </math>----------(3.3)</div> | |||
Here, <math>\varepsilon </math> is the molar extinction coefficient, 𝑙 is the optical path length, and 𝑐 is the solution concentration, 10mg/mL is the most commonly used concentration for film fabrication. Therefore, when the concentration and path length are kept as constant, changes in the UV-Vis spectrum mainly caused by solution aggregation. | |||
For conjugated polymers, aggregation changes the intermolecular interactions and electronic coupling between polymer chains. These changes can be seen in the UV-Vis spectrums, such as shifts in peak position, changes in peak shape, and differences in the relative intensity of characteristic absorption features. Therefore, by comparing UV-Vis spectrums under different solvents, temperatures, the degree of aggregation in solution and its changes can be evaluated. | |||
[[image:Fig3_2.png|700px|center]] | |||
<center> Fig 3.1. UV-Vis principal diagram. </center> | |||
The solution-state aggregation of conjugated polymers is governed by multiple factors, including molecular structure, molecular weight, solvent, temperature, concentration, and additives. Among these, solvent and temperature are two of the most commonly varied processing parameters and have the most direct influence on chain conformation and interchain interactions in solution. Solvent affects the balance between polymer-solvent and polymer-polymer interactions, thereby determining whether the polymer chains remain molecularly dispersed or form pre-aggregates. Temperature effects the extent and ordering of aggregation by changing chain mobility and aggregate stability. Therefore, in this work, solvent and temperature are selected as the key variables to investigate the solution-state aggregation behavior of the PBDB-T family polymers, before examining how such aggregation influences film formation and carrier transport. | |||
====3.1.2 Variable-solvent UV–Vis measurements in solution: The sandwiched method==== | |||
As shown in Fig 3.2, fixed temperature UV-Vis measurements were carried out on solutions prepared in different solvents in order to investigate the effect of solvent on solution-state aggregation, the setup shown in the figure. Two identical silicon substrates are selected and treated with oxygen plasma in order to improve surface wettability and ensure uniform spreading of the solution on the substrate surface. A piece of tape with a pre-cut hole in the center is taped tightly to one of the silicon substrates, after which an equal volume (40μL) of the test solution is deposited into the hole. The second silicon substrate is then placed on top as a cover. The two substrates are then tightly clamped along the edges with clips, which makes sure that solution has spread uniformly in the hole, and the optical path length of this part of solution is well-defined by the thickness of the tape and is consistent as per sample. After the sample is prepared, the solution UV-Vis was measured based on the principle shown in Fig. 3.1. | |||
[[image:Fig3_4.png|500px|center]] | |||
<center> Fig 3.2. Sample used for solution UV–Vis measurement by the sandwiched method: (a) top view and (b) side view.. </center> | |||
===3.2 J-V curve measurement=== | |||
====3.2.1 Mott-Gurney Law: The method to calculate carrier mobility==== | |||
In the SCLC regime, the current is mainly governed by bulk charge transport and follows the relation J∝V2. For a nearly trap-free unipolar device with efficient carrier injection, the current density can be described by the Mott–Gurney law: | |||
<div style="text-align: center;"> <math> J = \frac{9}{8} \varepsilon_0 \varepsilon_r \mu \frac{(V - V_{bi})^2}{d^3} </math>----------(3.4)</div> | |||
where <math>\varepsilon_0</math> is the vacuum permittivity, <math>\varepsilon_r</math> is the relative permittivity, <math>\mu </math> is the carrier mobility, Vbi is the built-in potential, and d is the thickness of the organic layer. Therefore, by fitting the J-V curve in the SCLC region using the Mott–Gurney law, the carrier mobility can be estimated. | |||
The applicability of the Mott-Gurney law requires unipolar transport, which means only one type of charge carrier dominates during transport; ohmic injection; and no electrical traps within the selected voltage range. By blocking electron injection and ensuring that the device current is dominated by holes, the HOD structure avoids the complication of electron–hole recombination. As a result, the measured current more accurately reflects hole transport in the organic layer, making the extraction of carrier mobility from the J-V characteristics more reliable. | |||
====3.2.2 Selection of device type==== | |||
Through UV–Vis experiments, we identified two factors that can affect solution aggregation: the solvent and halogen substitution. According to the study by Zhao et al., for annealed solutions, if spin-coating is carried out within a short period of time, the aggregation state in solution can be retained in the film state. As a result, the aggregation in these films differs from that of films prepared from solutions at room temperature. To further investigate the study’s objective, organic diodes were fabricated using solutions with different solvents and different annealing temperatures, and their J–V curves were measured. | |||
To enable the application of the Mott–Gurney law for quantifying carrier mobility, hole-only diodes (HODs) were fabricated. In this device structure, electron injection is effectively blocked, so the device current is dominated by holes. Such a structure obeys the using conditions of the Mott–Gurney law, in which the current raise from single-carrier, space-charge-limited transport in the bulk of the organic layer, instead of from bipolar conduction or injection-limited processes at the interfaces. As a result, hole transport becomes the dominant process, allowing the hole mobility to be extracted more reliably. Therefore, this structure provides a more straightforward and reliable way to compare the effects of different materials and solvents on charge transport. | |||
====3.2.3 Selection of hole injection layer and cathode material==== | |||
For the fabrication of hole-only diodes, the selection of the hole injection layer and cathode material is especially important. These two components determine the hole injection barrier and electron injection barrier, respectively, and therefore determine whether holes can be efficiently injected into the HOMO while electron injection is blocked by the electron injection barrier. | |||
A HIL can facilitate hole injection into the HOMO of the organic layer by tuning the energy-level alignment at the electrode/organic interface and increasing the effective work function of the anode. For the hole injection layer, para-CF₃-substituted poly(triarylamine-alt-fluorene)(pTFF) is used in this study. According to the research of Png R.-Q. et al., heavily p-doped pTFF exhibits an ultrahigh vacuum work function as 5.9eV. As a result, the deep work function can significantly reduce the hole injection barrier, and it can provide efficient hole injection into the organic semiconductor. Therefore, pTFF is highly suitable as the HIL in this study. | |||
[[image:Fig3_5.png|300px|center]] | |||
<center> Fig 3.3. Chemical structure of pTFF. </center> | |||
Among the metal materials commonly used as evaporated cathodes, Ag has a relatively deep work function, which helps increase the electron injection barrier and thereby effectively blocks electron injection. Therefore, Ag was chosen as the cathode in this study. | |||
====3.2.4 Processing of making organic diode==== | |||
In this study, the p-doping process of pTFF was as follows. First, pTFF was weighed and heated at 120°C for at least 1 h to fully remove residual moisture. It was then dissolved in acetonitrile (ACN) to prepare a 15 mg/mL pTFF solution. Separately, an appropriate amount of Magic Blue was weighed and dissolved in ACN to prepare a 12 mM dopant solution. The dopant solution was then mixed with the pTFF solution at a doping level of at least 0.8 eq. After that, dimethyl carbonate(DMC) with a volume of at least three times that of the mixed solution was added. he mixture was centrifuged, and the supernatant was removed. This washing process was repeated twice to remove Na+ and SbCl6−. Then, an appropriate amount of ACN was added to redisperse the sample to a concentration of 20 mM. DMC with a volume of at least three times that of the solution was again added, followed by centrifugation, precipitation, and removal of the supernatant. This process was also repeated twice. Finally, an appropriate amount of ACN was added to prepare a 15 mg/mL solution, completing the doping process. The doped solution was processed into a film of about 30 nm, and the doping level was determined by UV–Vis spectroscopy. The doping level should be above 0.8. A 30nm film was fabricated from doped solution, the spin-coating condition is 2000rpm, 40s. The doping level was measured by UV–Vis spectroscopy. | |||
During device fabrication, a p-doped pTFF solution was first prepared, and the spin-coating conditions were tested to obtain a uniform thin film. The pTFF film was then spin-coated, and its doping level and film thickness were measured. Before spin-coating film, the ITO substrates were pretreated as follows: The substrates were first marked and then cleaned with acetone and IPA, followed by oxygen plasma treatment for 10 mins to further remove residual contaminants and improve the surface condition. After these steps, the doped pTFF film was spin-coated onto the ITO substrates, and the film thickness was about 30 nm. | |||
During the fabrication of the organic semiconductor active layer, OSC films were spin-coated onto ITO/pTFF substrates under different conditions. In total, 11 groups of devices were fabricated, as summarized in the table below. The thickness of the OSC films was approximately 120 nm. Finally, Ag was deposited as the cathode by thermal evaporation to complete the device fabrication. | |||
[[image:Table3_1.png|500px|center]] | |||
[[image:Fig3_6.png|500px|center]] | |||
<center> Fig 3.4. Photograph of hole only organic diodes. </center> | |||
===3.3 Electro Absorption Spectroscopy=== | |||
====3.3.1 Theory==== | |||
Electro absorption technology is achieved by measuring the change in absorption coefficient after an applied electric field is applied[52][53][54][55]. Under normal incident conditions, the intensity I of light transmitted through the absorbing medium can be calculated using the Lambert-Beer law: | |||
<div style="text-align: center;"> <math> I = I_0 (1 - R)^2 e^{-\alpha d} </math>----------(3.5)</div> | |||
Where <math> I_0 </math> represents the original light intensity of the incident light, <math> R </math> is the reflectivity, <math> \alpha </math> is the absorption coefficient, and <math> d </math> is the thickness of the absorbing medium. | Where <math> I_0 </math> represents the original light intensity of the incident light, <math> R </math> is the reflectivity, <math> \alpha </math> is the absorption coefficient, and <math> d </math> is the thickness of the absorbing medium. | ||
| Line 19: | Line 155: | ||
Both <math> R </math> and <math> \alpha </math> are affected by the external electric field, and their <math> \Delta I </math> changes as follows: | Both <math> R </math> and <math> \alpha </math> are affected by the external electric field, and their <math> \Delta I </math> changes as follows: | ||
<div style="text-align: center;"> <math> \Delta I = -I \left( \frac{2R}{1 - R} \frac{\Delta R}{R} + d \Delta \alpha \right) </math>----------( | <div style="text-align: center;"> <math> \Delta I = -I \left( \frac{2R}{1 - R} \frac{\Delta R}{R} + d \Delta \alpha \right) </math>----------(3.6)</div> | ||
Under classical operating conditions, the change in the value of <math> R </math> is negligible. Equation ( | Under classical operating conditions, the change in the value of <math> R </math> is negligible. Equation (3.6) simplifies to: | ||
<div style="text-align: center;"> <math> \frac{\Delta I}{I} = -d \Delta \alpha </math>----------(3)</div> | <div style="text-align: center;"> <math> \frac{\Delta I}{I} = -d \Delta \alpha </math>----------(3.7)</div> | ||
From a microscopic perspective, the energy level E(F) of state <math>\langle i |</math> in electric field F is given by the following equation: | From a microscopic perspective, the energy level E(F) of state <math>\langle i |</math> in electric field F is given by the following equation: | ||
<div style="text-align: center;"> <math> E(F) = E(0) - m_i F - \frac{1}{2}(p_i F) F </math>----------( | <div style="text-align: center;"> <math> E(F) = E(0) - m_i F - \frac{1}{2}(p_i F) F </math>----------(3.8)</div> | ||
<math>m_i</math> represents the electric dipole moment of this state, and <math>p_i</math> is its polarizability. Therefore, the optical transition energy shift <math>\Delta E</math> from the initial state to the final state is given by the following equation: | <math>m_i</math> represents the electric dipole moment of this state, and <math>p_i</math> is its polarizability. Therefore, the optical transition energy shift <math>\Delta E</math> from the initial state to the final state is given by the following equation: | ||
<div style="text-align: center;"> <math> \Delta E(F) = -(m_f - m_i)F - \frac{1}{2}(p_f - p_i)F^2 </math>----------( | <div style="text-align: center;"> <math> \Delta E(F) = -(m_f - m_i)F - \frac{1}{2}(p_f - p_i)F^2 </math>----------(3.9)</div> | ||
The first term represents the linear energy change caused by the difference in dipole moments between the initial and final states, which cancels out in isotropic solids without permanent dipoles. The second term describes the energy shift caused by the difference in polarizability between the initial and final states, an effect that always exists. | The first term represents the linear energy change caused by the difference in dipole moments between the initial and final states, which cancels out in isotropic solids without permanent dipoles. The second term describes the energy shift caused by the difference in polarizability between the initial and final states, an effect that always exists. | ||
| Line 37: | Line 173: | ||
The absorption variation of the external electric field <math>\Delta \alpha</math> can be represented by the <math>\Delta E</math> term of the Maclaurin series, while the third and higher order terms can be ignored due to the small spectral changes. | The absorption variation of the external electric field <math>\Delta \alpha</math> can be represented by the <math>\Delta E</math> term of the Maclaurin series, while the third and higher order terms can be ignored due to the small spectral changes. | ||
<div style="text-align: center;"> <math> \Delta \alpha(h\nu) = \left( \frac{d\alpha}{dE}\Delta E \right) + \frac{1}{2}\left( \frac{d^2\alpha}{d^2 E}\Delta E^2 \right)</math>----------( | <div style="text-align: center;"> <math> \Delta \alpha(h\nu) = \left( \frac{d\alpha}{dE}\Delta E \right) + \frac{1}{2}\left( \frac{d^2\alpha}{d^2 E}\Delta E^2 \right)</math>----------(3.10)</div> | ||
The sole contribution of the first term comes from the second-order Strac effect <math>\frac{1}{2} \Delta p F^2</math>, while the isotropic average of <math>(\Delta m F)^2</math> in the second term does not cancel out, but instead produces <math>\frac{1}{3} (\Delta m F)^2</math> in the randomly distributed matrix. The third contribution of <math>\Delta \alpha (hv) </math> comes from the transition of the oscillator to the previously forbidden state when the electric field is present, and this transition also has a quadratic relationship with F. | The sole contribution of the first term comes from the second-order Strac effect <math>\frac{1}{2} \Delta p F^2</math>, while the isotropic average of <math>(\Delta m F)^2</math> in the second term does not cancel out, but instead produces <math>\frac{1}{3} (\Delta m F)^2</math> in the randomly distributed matrix. The third contribution of <math>\Delta \alpha (hv) </math> comes from the transition of the oscillator to the previously forbidden state when the electric field is present, and this transition also has a quadratic relationship with F. | ||
| Line 43: | Line 179: | ||
Adding up all contributions to <math>\Delta \alpha (hv) </math>, we get: | Adding up all contributions to <math>\Delta \alpha (hv) </math>, we get: | ||
<div style="text-align: center;"> <math> \Delta \alpha = \left[ a \alpha + b \frac{d\alpha}{dE} + C \frac{d^2 \alpha}{d^2 E} \right] F^2</math>----------( | <div style="text-align: center;"> <math> \Delta \alpha = \left[ a \alpha + b \frac{d\alpha}{dE} + C \frac{d^2 \alpha}{d^2 E} \right] F^2</math>----------(3.11)</div> | ||
For a composite electric field with AC component <math>E_{ac} \sin(\omega t)</math> and DC component <math>E_dc</math>: | |||
<div style="text-align: center;"> <math> E(t) = E_{ac} \sin(\omega t) + E_dc </math>----------(3.12)</div> | |||
Substituting formula (3.12) into (3.11) yields the modulation of <math> \Delta \alpha </math> at the fundamental frequencies <math> 1\omega </math> and <math> 2\omega </math>: | |||
<div style="text-align: center;"> <math> \Delta \alpha \propto \left[ \frac{1}{2} E_{ac}^2 \left( 1 + \sin\left( 2\omega t - \frac{\pi}{4} \right) \right) + 2E_{ac}(E_{dc} - E_{bi})\sin(\omega t) + (E_{dc} - E_{bi})^2 \right]</math>----------(3.13)</div> | |||
<div style="text-align: center;"> <math>E_{bi} = \frac{\phi_{anode} - \phi_{cathode}}{qd} </math>----------(3.14)</div> | |||
where <math>\phi_{anode}</math> and <math>\phi_{cathode}</math> are the work functions of the two electrodes, q is the elementary charge, and d is the film thickness. | |||
Using phase-sensitive lock-in detection, the harmonic components can be separated. Since ∆α(1ω) varies linearly with <math> E_{dc}-E_{bi} </math>, the built-in field can be obtained by identifying the DC bias at which the 1ω electro absorption signal disappears. In this way, electro absorption spectroscopy provides a direct method for determining the built-in potential of the diode. | |||
====3.3.2 EA device setup and operation procedures==== | |||
Before the EA measurement, the diode device was first connected to the cryostat inside the glovebox. The cryostat was then fixed on the holder, and the connection of the device was verified by measuring the J–V curve. After that, the cryostat was evacuated to 10−6mbar. Once the required pressure was reached, a pre-measurement was carried out to further confirm that the connection was normal, after which the system was cooled down. When the temperature inside the chamber reached 30 K, the formal EA measurement was performed. | |||
[[image:Fig3_7.png|400px|center]] | |||
<center> Fig 3.5. Schematic diagram of the EA setup </center> | |||
[[image:Fig3_8.png|200px|center]] | |||
<center> Fig 3.6. Photograph of the device in cryostat </center> | |||
As shown in the schematic of the setup, direct measurement of the transmission signal is difficult due to the device structure. Therefore, in this experiment, the transmission signal was inferred from the measured reflection signal ∆R. | |||
This device is driven by a modulated driving voltage. The DC bias voltage varies from 0V to 3V in 0.5V steps to change the electric field strength, while a constant AC bias voltage is applied simultaneously. The change in electric field strength after applying this driving voltage leads to a change in the absorption bandgap ΔE. | |||
Monochromatic light is incident on a glass substrate at a 45° angle, and the reflected light illuminates a photodiode. The voltage output of the photodiode is demodulated by a lock-in amplifier. This voltage output reflects the change in reflectivity with the excitation bias voltage Vdc within a certain photon energy range. | |||
====3.3.3 Method for calculating carrier mobility ==== | |||
By comparing the Vbi values of the CB solution system at RT and 60°C, it can be inferred that the Vbi at 100°C is close to the theoretical value of 1.2 V. Therefore, EA measurement for the 100°C devices is not necessary. After obtaining the specific Vbi of the device, the carrier mobility can be quantified using the Mott–Gurney law. | |||
Although the Mott–Gurney law can directly provide an equation for carrier mobility, its application requires several ideal conditions, such as a uniform electric field, trap-free transport, and current dominated by SCLC. | |||
Pasveer demonstrated that in disordered organic semiconductors, the carrier mobility μis dependent on both the local carrier density p and the electric field E. Neglecting these dependences may lead to an inaccurate estimation of carrier mobility. Therefore, a modified form of the Mott–Gurney law is required to obtain a more reliable value of carrier mobility. In this study, the derivation was carried out based on Gauss’s law and the drift–diffusion equation to better describe the physical processes in the device and obtain more reliable mobility values. The derivation proceeds as follows: | |||
Gauss’s Law: <math>\nabla \cdot E = \rho e / \varepsilon</math>, in 1D: | |||
<div style="text-align: center;"> <math>\frac{dE}{dd} = \frac{\rho e}{\varepsilon}</math>----------(3.15)</div> | |||
MG equation: | |||
<div style="text-align: center;"> <math>J = \frac{9}{8} \varepsilon \mu \frac{V^2}{d^3}</math>----------(3.16)</div> | |||
Drift-diffusion equation: | |||
<div style="text-align: center;"> <math>J = ep\mu E - De \frac{dp}{d\text{d}} = \epsilon\mu E \frac{dE}{d\text{d}} - \epsilon D \frac{d^{2}E}{d\text{d}^{2}} </math>----------(3.17)</div> | |||
Integrate, we can get | |||
<div style="text-align: center;"> <math>J\text{d} = \frac{1}{2} \epsilon \mu E^{2} - \epsilon D \frac{dE}{d\text{d}}</math>----------(3.18)</div> | |||
Neglecting the <math>dE/dd </math> term since we assume constant electrical field across OSC, then | |||
<div style="text-align: center;"> <math>E(z) = \sqrt{\frac{2J}{\varepsilon \mu}} \sqrt{d}</math>----------(3.19)</div> | |||
<div style="text-align: center;"> <math>\langle E \rangle = \frac{1}{d} \int_0^Z E(d)dd = \frac{1}{d} \int_0^Z \sqrt{\frac{2J}{\varepsilon \mu}} \sqrt{d} dd = \frac{1}{d} \cdot \frac{3}{2} \cdot \frac{2}{3} d^{\frac{3}{2}} \cdot \sqrt{\frac{V^2}{d^3}} = \frac{V}{d}</math>----------(3.20)</div> | |||
From Gauss’s Law, we can calculate the carrier density: | |||
<div style="text-align: center;"> <math>p(d) = \frac{d}{dd} \left( \sqrt{\frac{2J}{\varepsilon \mu}} \sqrt{d} \right) \cdot \frac{\varepsilon}{e} = \frac{1}{e} \sqrt{\frac{\varepsilon J}{2\mu}} \frac{1}{\sqrt{d}}</math>----------(3.21)</div> | |||
The average carrier density is: | |||
<div style="text-align: center;"> <math> \langle p \rangle = \frac{1}{d} \int_0^Z p(d) dd = \frac{1}{d} \int_0^d \frac{1}{e} \sqrt{\frac{\varepsilon J}{2\mu}} \frac{1}{\sqrt{d}} dd = \frac{1}{d} \cdot \frac{1}{e} \cdot 2\sqrt{d} \sqrt{\frac{\varepsilon J}{2\mu}} = \frac{1}{e} \frac{3}{2} \cdot \left( \frac{\varepsilon}{d} \right) \frac{V}{d}</math>----------(3.22)</div> | |||
In Mott-Gurney regime: | |||
<div style="text-align: center;"> <math> \langle Ep \rangle = \frac{1}{d} \int_0^d E(d)p(d) dd = \frac{1}{de} \int_0^d \sqrt{\frac{\varepsilon J}{2\mu}} \frac{1}{\sqrt{d}} \cdot \sqrt{\frac{2J}{\varepsilon \mu}} \cdot \sqrt{d} dd = \frac{1}{e} \frac{J}{\mu} = \frac{3}{4} \langle E \rangle \langle p \rangle</math>----------(3.23)</div> | |||
<div style="text-align: center;"> <math> \langle \mu \rangle = \frac{J}{e \langle Ep \rangle} = \frac{J}{3/4 \cdot \langle p \rangle \langle E \rangle e} </math>----------(3.24)</div> | |||
==4. Results and discussion== | |||
===4.1 Results of UV-Vis=== | |||
====4.1.1 Result of variable-solvent==== | |||
[[image:Fig4_2.png|700px|center]] | |||
<center> Fig4.1 UV–Vis spectra of (a) PM6 in CB and PM6 in CF, (b) PBDB-T in CB and PBDB-T in CF, measured at a solution concentration of 10mg/mL using the sandwiched method. </center> | |||
For both polymers, the difference between the two characteristic peaks is larger in CB than in CF. This suggests that the polymer chains show a stronger aggregation tendency in CB under the same conditions. The same trend is observed for both PM6 and PBDB-T, indicating that CB leads to a higher degree of solution-state aggregation than CF in these two polymer systems. In addition, no clear change is observed between the spectrums recorded at 0 min and 60 min for any of the four solutions. This indicates that all four solutions remain stable over the measured time range, and their aggregation states do not change significantly with time. | |||
====4.1.2 Conclusion for UV-Vis measurement==== | |||
UV–Vis measurements were used to test the solution-state aggregation behavior of the PBDB-T family polymers and to evaluate the effects of solvent and temperature on their aggregation in solution. Under the same conditions, PM6 and PBDB-T show a stronger aggregation in CB than in CF. These results show that UV–Vis measurements can reveal the solution pre-aggregation state under different processing conditions before film deposition. Zhao[4] mentions that for annealed solutions, if spin-coating is carried out within a short period of time, the aggregation state in solution can be retained in the film state. So, the results further indicate that the initial aggregation state during film formation can be tuned by solvent selection and solution-heating conditions, thereby influencing the aggregation and microstructure of the resulting films. | |||
===4.2 Result of J-V curve=== | |||
[[image:Fig4_3.png|700px|center]] | |||
<center> Fig4.2 J–V curves of (a) PM6 and (b) PBDB-T devices processed from CB and CF at RT and 60 °C. Different line styles represent different pixels of the same device, and the vertical line marks 2V. </center> | |||
Since the forward and reverse scan curves are very similar, only the forward scan curves are shown in order to get clear results. | |||
According to the Mott–Gurney law, when the film thickness and applied voltage are the same, the carrier mobility μ is proportional to the current density J. Therefore, carrier mobility can be estimated by comparing the current density of films with the same thickness at the same voltage. | |||
As shown in Fig. 4.2, at the same temperature, the mobility of PM6 films processed from CB is higher than that of films processed from CF, while the opposite trend is observed for PBDB-T. These results indicate that solvent processing conditions can significantly influence the carrier mobility of the films. | |||
Because the above conclusion was reached under the assumption that different devices have the same film thickness and built-in potential, it may be inaccurate if the actual device thickness or built-in potential varies. Therefore, electro absorption(EA) spectroscopy is required to determine <math>V_{bi} </math> of each device, so that carrier mobility can be quantified using the Mott-Gurney law. | |||
===4.3 Result of EA === | |||
From the theory of EA, <math>V_{bi} </math> of different device can be measured. According to EA theory, the null bias is the DC bias that cancels the built-in electric field in the device. At this bias, the Stark shift becomes zero, so this voltage is taken as <math>V_{bi} </math>. In this study, as <math>V_{dc} </math> increases, the EA feature changes from a peak to a dip, which happens near 1.85eV. The null bias is taken as the voltage between these two cases, where the feature becomes almost flat. This voltage is defined as the built-in potential <math>V_{bi} </math>. Here is the result of EA: | |||
[[image:Fig4_3(ab).png|700px|center]] | |||
[[image:Fig4_3(cd).png|700px|center]] | |||
<center> Fig4.3 (a) EA spectrum of PM6 in CB RT. (b) EA spectrum of PBDB-T in CB RT. (c) EA spectrum of PM6 in CF RT. (d) EA spectrum of PBDB-T in CF RT. </center> | |||
[[image:Table4_1.png|700px|center]] | |||
===4.4 Result of carrier mobility=== | |||
Based on the above equations, the carrier mobility was calculated using the J–V curve data, and the results are shown below. | |||
[[image:Fig4_4.png|700px|center]] | |||
<center> Fig4.4 (a) Carrier mobility of PM6: films with thickness ranging from 120.6 to128.8nm processed from CB and CF. (b) Carrier mobility of PBDB-T: films with thickness ranging from 125.5 to 149.5nm processed from CB and CF. </center> | |||
[[image:Table4_2.png|700px|center]] | |||
As shown in Figure 4.4, solvent type has a significant influence on charge carrier mobility. For films of the same polymer measured under similar conditions, the mobility of the CB-based film is much higher than that of the CF-based film. This result indicates that solvent processing can change the solution-state aggregation and thereby affect charge transport. For both PBDB-T and PM6, CB is a more suitable solvent than CF. In the CB system, both polymers show better solubility and stronger aggregation in solution, and the corresponding films exhibit higher carrier mobility. These results suggest that, within the processing range studied here, solvent-dependent aggregation plays a key role in determining charge-transport behavior. | |||
==Conclusion== | |||
This study systematically investigates how solvent type affects the aggregation behavior and charge transport of PBDB-T family polymers, and further explores how solvent-controlled aggregation influences molecular packing and film morphology. | |||
The J–V curves and the extracted carrier mobility show that solvent type strongly affects charge transport. The experimental results demonstrate that, among the solvent-processing parameters studied here, solvent type plays the dominant role in determining carrier mobility. In particular, films prepared from CB exhibit much higher carrier mobility than those prepared from CF. By relating solution-state aggregation to carrier mobility, the results further suggest that solvent-dependent aggregation plays a critical role in charge transport. | |||
A possible explanation is that CB promotes a more favorable aggregation state in solution, which provides a certain degree of molecular pre-organization before or during film formation. This pre-organization helps establish more ordered π–π stacking, enhances intermolecular electron cloud overlap, and enables more continuous charge-transport pathways, thereby leading to higher carrier mobility. In contrast, CF leads to a less favorable aggregation state, which is less effective in supporting ordered molecular packing and continuous transport pathways. | |||
Overall, this study shows that charge transport in organic semiconductors can be strongly influenced by solvent type. The effect of solvent type is not limited to the solution state, but extends to molecular packing, film morphology, and the continuity of charge-transport pathways in the solid film. Therefore, controlling aggregation through solvent selection provides an effective strategy for improving charge transport in PBDB-T family polymer systems. | |||
==References== | |||
[1].Koh, Q. M., Mazlan, N. S., Seah, Q. J., Yang, J. C., Chen, Y. J., Png, R. Q., Ho, P. K. H. & Chua, L. L., ACS Appl Mater Interfaces 2024, 16, 39708-39716. | |||
[2].Sachnik, O., Tan, X., Dou, D., Haese, C., Kinaret, N., Lin, K. H., Andrienko, D., Baumgarten, M., Graf, R., Wetzelaer, G. A. H., Michels, J. J. & Blom, P. W. M., Nat Mater 2023, 22, 1114-1120. | |||
[3].Ang, Adele A.H., Xiong, X.C., Yang, J.C., Hong, Z.L., Png, R.Q., Chua, L.L., Ho, Peter K.H. (unpublished). | |||
[4].Zhao, L. H., Ping, R. Q., Zhuo, J. M., Wong, L. Y., Tang, J. C., Su, Y. S. & Chua, L. L., Macromolecules 2011, 44, 9692–9702. | |||
[5].Li, Y., Yan, S., Geng, M. & Li, H., Enhancing π–π stacking by a halogen substituent in a single-molecule junction, Chem. Commun. 2025, 61, 8419–8422. | |||
[6].Sirringhaus, H., Tessler, N. & Friend, R. H., Integrated optoelectronic devices based on conjugated polymers, Science 1998, 280, 1741–1744. | |||
[7].Facchetti, A., π-Conjugated polymers for organic electronics and photovoltaic cell applications, Chem. Mater. 2011, 23, 733–758. | |||
The | [8].Forrest, S. R., The path to ubiquitous and low-cost organic electronic appliances on plastic, Nature 2004, 428, 911–918. | ||
[9].Sirringhaus, H., Device physics of solution-processed organic field-effect transistors, Adv. Mater. 2005, 17, 2411–2425. | |||
[10].Zhang, S., Qin, Y., Zhu, J. & Hou, J., Over 14% efficiency in polymer solar cells enabled by a chlorinated polymer donor, Adv. Mater. 2018, 30, 1800868. | |||
[11].Hou, J., Inganäs, O., Friend, R. H. & Gao, F., Organic solar cells based on non-fullerene acceptors, Nat. Mater. 2018, 17, 119–128. | |||
[12].Facchetti, A., π-Conjugated polymers for organic electronics and photovoltaic cell applications, Chem. Mater. 2011, 23, 733–758. | |||
[13].Blom, P. W. M. & de Jong, M. J. M., Electrical characterization of organic semiconductor devices, IEEE J. Sel. Top. Quantum Electron. 2000, 6, 1059–1068. | |||
[14].Brütting, W., Adachi, C., Physics of Organic Semiconductors, Wiley-VCH, 2012. | |||
[15].Zhou, M., Energy Level Alignment in Semiconducting Organic Electronic Devices, Ph.D. Dissertation, National University of Singapore, Singapore, 2010. | |||
[16].Blom, P. W. M. & de Jong, M. J. M., Electrical characterization of organic semiconductor devices, IEEE J. Sel. Top. Quantum Electron. 2000, 6, 1059–1068. | |||
[17].Mott, N. F. & Gurney, R. W., Electronic Processes in Ionic Crystals, Oxford University Press, Oxford, 1940. | |||
[18].Zhou, M., Chua, L.-L., Png, R.-Q., Yong, C.-K., Sivaramakrishnan, S., Chia, P.-J., Wee, A. T. S., Friend, R. H. & Ho, P. K. H., Hole-Doped Interfaces at Ohmic Contacts to Organic Semiconductors, Phys. Rev. Lett. 2009, 103, 036601. | |||
[19].Haneef, H. F., Zeidell, A. M. & Jurchescu, O. D., Charge Carrier Traps in Organic Semiconductors: A Review on the Underlying Physics and Impact on Electronic Devices, J. Mater. Chem. C 2020, 8, 759–787. | |||
[20].Liu, Z. & Lin, Y., Testing Trap States in Polymer Solar Cells, Polym. Test. 2024, 108387. | |||
[21].Zhao, C., Tang, C. G., Seah, Z.-L., Koh, Q.-M., Chua, L.-L., Png, R.-Q. & Ho, P. K. H., Improving organic photovoltaic cells by forcing electrode work function well beyond onset of Ohmic transition, Nat. Commun. 2021, 12, 2358. | |||
[22].Urbančič, J., Charge Transport in Organic Semiconductors, IntechOpen, 2025. | |||
[23].Zhao, N., Osman, K., Png, R. Q., Ho, P. K. H. & Chua, L.-L., Effect of side-chain length on the structure and charge transport in conjugated polymers, J. Am. Chem. Soc. 2010, 132, 15362–15363. | |||
[24].Aasmundtveit, K. E.; Samuelsen, E. J.; Guldstein, M.; Steinsland, C.; Flornes, O.; Fagermo, C.; Seeberg, T. M.; Pettersson, L. A. A.; Inganäs, O.; Feidenhans’l, R.; Ferrer, S. Macromolecules 2000, 33, 3120-3127. | |||
[25].Chang, J. F.; Sun, B.; Breiby, D. W.; Nielsen, M. M.; Sölling, T. I.; Giles, M.; McCulloch, I.; Sirringhaus, H. Chem. Mater. 2004, 16, 4772-4776. | |||
[26].Zhao, L.-H.; Png, R.-Q.; Zhuo, J.-M.; Wong, L.-Y.; Tang, J.-C.; Su, Y.-S.; Chua, L.-L. Role of Borderline Solvents to Induce Pronounced Extended-Chain Lamellar Order in π-Stackable Polymers. Macromolecules 2011, 44 (24), 9692–9702. | |||
[27].Chang, J. F.; Clark, J.; Zhao, N.; Sirringhaus, H.; Breiby, D. W.; Andreasen, J. W.; Nielsen, M. M.; Giles, M.; Heeney, M.; McCulloch, I. Phys. Rev. B 2006, 74, 115318-1-12. | |||
[28].Samitsu, S.; Shimomura, T.; Heike, S.; Hashizume, T.; Ito, K. Macromolecules 2008, 41, 8000-8010. | |||
[29].Kim, D. H.; Park, Y. D.; Jang, Y.; Kim, S.; Cho, K. Macromol. Rapid Commun. 2005, 26, 834-839. | |||
[30].Merlo, J. A.; Frisbie, C. D. J. Polym. Sci., B: Polym. Phys. 2003, 41, 2674-2680. | |||
[31].Wang, S.; Tang, J. C.; Zhao, L. H.; Png, R. Q.; Wong, L. Y.; Chia, P. J.; Chan, H. S. O.; Ho, P. K. H.; Chua, L. L. Appl. Phys. Lett. 2008, 93, 162103-1-3. | |||
[32].Sperling, L. H. Introduction to Physical Polymer Science, 3rd ed.; Wiley-Interscience: New York, 2001. | |||
[33].Tan, Z.-S., Jamal, Z., Teo, D. W. Y., Ko, H.-C., Seah, Z.-L., Phua, H.-Y., Ho, P. K. H., Png, R.-Q. & Chua, L.-L., Optimization of fluorinated phenyl azides as universal photocrosslinkers for semiconducting polymers, Nat. Commun. 2024, 15, 6354. | |||
[34].Png, R.-Q., Chia, P.-J., Tang, J.-C., Liu, B., Sivaramakrishnan, S., Zhou, M., Khong, S.-H., Chan, H. S. O., Burroughes, J. H., Chua, L.-L., Friend, R. H. & Ho, P. K. H., High-performance polymer semiconducting heterostructure devices by nitrene-mediated photocrosslinking of alkyl side chains, Nat. Mater. 2010, 9, 152–158. | |||
[35].Zhang, Q., Kelly, M. A., Bauer, N. & You, W., The Curious Case of Fluorination of Conjugated Polymers for Solar Cells, Acc. Chem. Res. 2017, 50, 2401–2409. | |||
[36].Wheeler, S. E., Understanding Substituent Effects in Noncovalent Interactions Involving Aromatic Rings, Acc. Chem. Res. 2013, 46, 1029–1038. | |||
[37].Kim, I.-B., Jang, S.-Y., Kim, Y.-A., Kang, R., Kim, I.-S., Ko, D.-K. & Kim, D.-Y., The Effect of Fluorine Substitution on the Molecular Interactions and Performance in Polymer Solar Cells, ACS Appl. Mater. Interfaces 2017, 9, 24011–24019. | |||
[38].Leclerc, N., Chávez, P., Ibraikulov, O. A., Heiser, T. & Lévêque, P., Impact of Backbone Fluorination on π-Conjugated Polymers in Organic Photovoltaic Devices: A Review, Polymers 2016, 8, 11. | |||
[39].Zheng, Z., Yao, H., Ye, L., Xu, Y., Zhang, S. & Hou, J., PBDB-T and its Derivatives: A Family of Polymer Donors Enables over 17% Efficiency in Organic Photovoltaics, Mater. Today 2020, 35, 115–130. | |||
[40].Grüne, J., Stähly, B., Lulei, S., Kotova, M., Dyakonov, V., Gillett, A. J. & Sperlich, A., Triplet Excitons and Associated Efficiency-Limiting Pathways in Organic Solar Cell Blends Based on Non-Halogenated PBDB-T and Y-Series, Fluorinated PM6:Y6, and Chlorinated PM7:Y7, Adv. Funct. Mater. 2023, 33, 2212640. | |||
Latest revision as of 14:51, 9 April 2026
Team members[edit | edit source]
Li Jinhan A0327554Y
Liu Chenyang A0328377R
1. Introduction[edit | edit source]
1.1 Fundamentals of polymer organic semiconductors[edit | edit source]
Organic semiconductors are a class of materials composed of organic molecules or polymers that possess semiconductor-like energy levels and are capable of transporting electrical charges. Compared with inorganic semiconductors, organic semiconductors offer advantages such as structural tunability, solution processability, and compatibility with flexible electronic devices[8]. Among them, polymer are particularly important because their long-chain molecular structures enable good film-forming ability and make them highly suitable for large-area solution processing.
PBDB-T is a representative polymer that has attracted significant attention due to its excellent optoelectronic properties and widespread use in organic electronic devices, especially organic solar cells. Structurally, PBDB-T consists of repeating molecular units that form a conjugated polymer backbone. The conjugated backbone plays a central role in determining the semiconducting behavior of the material, as it governs the energy levels and facilitates charge transport along the polymer chain. In addition, PBDB-T contains alkyl side chains attached to the backbone. These side chains mainly improve the solubility and processability of the polymer, which are essential for solution-based fabrication methods. Therefore, the molecular structure of PBDB-T, including its repeating units, conjugated backbone, and side chains, provides the basis for understanding its aggregation behavior, film formation, and charge-transport properties.
1.2 Basic device for organic semiconductor research[edit | edit source]
In studies of organic semiconductors, organic diodes are fundamental devices for investigating charge-transport behavior. Typically, an organic diode consists of an ITO anode, a metal cathode, and an organic semiconductor thin film sandwiched between the two electrodes.
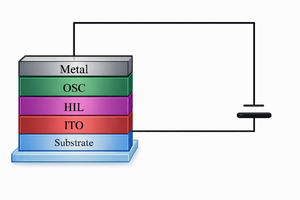
To achieve the highest efficiency, we usually utilize hole injection layer and electron injection layer to make sure of the efficient charge injection at both hole and electron contacts, which is guaranteed by the match of the energy levels of these various layers.
The work function is the minimum energy required to move an electron from the Fermi level to the vacuum level. The injection barrier is the energy difference that carriers must overcome in order to be injected from an electrode into the organic semiconductor. Meanwhile, in organic semiconductor diodes, an Ohmic contact refers to a contact at which the carrier injection and collection resistance at the metal/organic-semiconductor interface is lower than the bulk transport resistance of the organic semiconductor, so that device operation is no longer contact-limited but primarily bulk-transport-limited. In an organic semiconductor device, a built-in potential, Vbi, is formed even at zero applied bias because of the difference in the work functions of the two electrodes and the interfacial energy-level alignment. This built-in potential plays a central role in determining the internal electric field distribution and carrier injection behavior, and is therefore an important parameter for understanding charge transport in organic diodes.
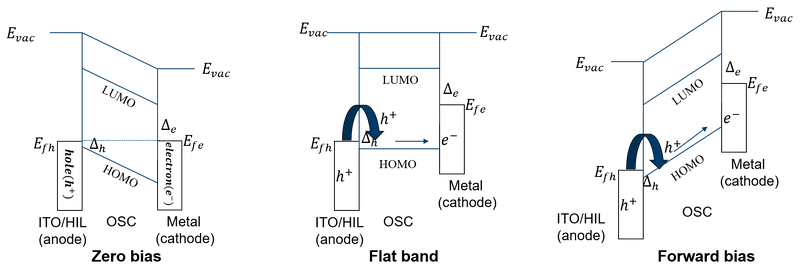
More importantly, the energy-level diagram shows the energy-level matching in the diode. It refers to the alignment between work function of the electrodes and the frontier energy levels of the organic semiconductor, as well as the resulting interfacial energy-level alignment. Such alignment determines the carrier injection and extraction barriers at the contacts. It effects the interfacial resistance and indicates whether the device is contact-limited or forms the Ohmic-contact. In general, better energy-level matching leads to smaller carrier-injection barriers and lower interfacial resistance. It also promotes Ohmic contact formation. In addition, the energy-level matching determines the built-in potential, and thus influences the internal electric field, charge transport, and overall device performance.
At the same time, the energy-level diagram also reveals the operating principle of the organic diode. At zero bias, the Fermi levels of the anode and cathode are aligned. Because the two electrodes have different work functions, a built-in potential is formed, and creates an internal electric field directed from the cathode to the anode. Meanwhile, hole and electron injection are limited by their injection barriers, so carrier injection is weak and the current remains low. As the applied bias increases and compensates the built-in potential, the device reaches the flat-band condition. In this state, the energy-level bending in the organic layer disappears, and the net internal electric field becomes zero. Therefore, this condition is commonly used to characterize the built-in potential, Vbi. When the applied bias further exceeds Vbi, the device enters a clear forward-conduction regime. The energy levels then tilt further. The forward bias changes internal electric field, making it easier for carriers to be injected into the HOMO or LUMO and thereby promoting charge transport through the device. Under this condition, if the electron (hole) injection barrier is sufficiently large while the hole (electron) injection barrier is small, electron (hole) injection is effectively blocked, while holes (electrons) can be injected efficiently, resulting in unipolar diode.
During the experiment, carrier transport is investigated by measuring the current density-voltage (J-V) characteristics of the diode. The J-V curve is a basic but powerful tool. It provides direct information about the charge-transport behavior in the organic semiconductor layer. At low bias, the device usually operates in the Ohmic regime, where the current increases approximately linearly with voltage. As the applied voltage increases, however, more carriers are injected into the organic layer and begin to accumulate. This accumulation forms space charge. The space charge distorts the internal electric field and limits the current, causing the device to enter the space-charge-limited current (SCLC) regime. In this regime, the current is mainly controlled by bulk transport in the organic semiconductor. Charge transport may also be affected by traps. A trap is a localized energy level within the bandgap that captures and confines charge carriers, thereby reducing their effective transport. One important feature in the J-V curve is the knee point. This point marks the transition from the Ohmic regime to the SCLC regime as the voltage increases. Therefore, by analyzing the shape of the J-V curve and the position of the knee point, it is possible to evaluate carrier transport and to identify the influence of space charge and trap effects in the device.
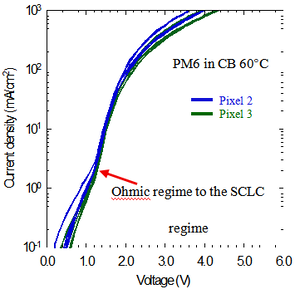
In this study, carrier transport behavior is quantitatively evaluated in terms of carrier mobility. Mobility describes how well charge carriers move under an electric field, and it is one of the most important parameters for characterizing charge transport in organic semiconductors. A higher mobility generally indicates more efficient carrier transport within the material. Carrier mobility is influenced by multiple factors. These include backbone planarity, aggregation, side-chain structure, and other molecular or microstructural features of the polymer semiconductor. Among these factors, aggregation is particularly important because it is closely related to molecular packing and charge-transport pathways. Aggregation refers to the tendency of polymer chains, either in solution or in a film, to associate with one another rather than remain fully separated. As a result, small clusters or more ordered structures can be formed[4]. The aggregation state in solution strongly affects the film-formation process and can further influence the final charge-transport properties, including mobility. Aggregation itself can be affected by factors such as solution processing and chemical substitution. Therefore, this study mainly focuses on how aggregation influences carrier mobility, with particular attention paid to the effects of solvent processing and substitution on the aggregation behavior of the polymer system. Since aggregation plays an important role in determining carrier mobility, it is necessary to further understand the factors that govern aggregation behavior in polymer semiconductors. In this work, two key factors are considered: solvent processing and halogen substitution. These two factors influence aggregation from different perspectives. Solvent processing mainly affects the solution-state behavior of polymer chains, whereas halogen substitution changes the intrinsic molecular interactions of the polymer backbone. In the following sections, these two effects will be discussed in detail.
1.3 Solvent[edit | edit source]
For polymer systems, solvents are generally classified as either good solvents or poor solvents. In a good solvent, strong polymer-solvent interactions enable effective solvation of the polymer chains, such that the chains adopt relatively extended conformations and remain well dispersed, with only a weak tendency toward aggregation. By contrast, in a poor solvent, polymer-solvent interactions are comparatively weak, while interchain interactions become more significant. Under these conditions, the polymer chains tend to contract, entangle, and aggregate, and may eventually precipitate from solution.
For conjugated polymers, however, this binary classification is often insufficient to describe their solution behavior in a comprehensive manner. Zhao et al. introduced the concept of a borderline solvent, defined as a solvent with a solvating ability intermediate between that of a good solvent and a poor solvent. In such a medium, the polymer chains are not fully extended and uniformly dispersed, as would be expected in a good solvent. Instead, they are more prone to intermolecular approach and the formation of a certain degree of ordered aggregation, while remaining dispersed in solution without obvious precipitation. Borderline solvents therefore have particular significance in the regulation of aggregation in conjugated polymer systems, and consequently exert an important influence on subsequent film formation, thin-film morphology, and device performance.
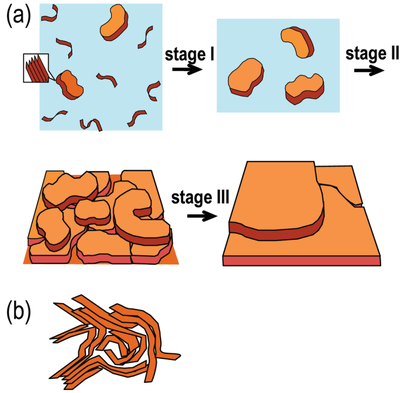
As shown in Figure 1.5, Zhao et al. proposed a mechanism by which a borderline solvent can induce ordered aggregation in conjugated polymers. In a borderline solvent, the polymer chains are more likely to undergo an early transition from coil-like conformations to more extended chain conformations, and subsequently form small, ordered 𝜋-stacking aggregates. These initial aggregates can then further organize and grow into more distinct plate-like stacked structures with a high degree of order and a preferred orientation. During the subsequent spin-coating process, the aggregates continue to spread, develop, and align on the substrate, thereby promoting the formation of an ordered thin film. Zhao et al. suggested that such dilute-solution 𝜋-stacking and the resulting extended-chain aggregates are crucial for achieving pronounced lamellar order in conjugated polymer films.
By contrast, in a good solvent, polymer–solvent interactions are sufficiently strong to maintain the polymer chains in a more fully solvated and dispersed state. Under these conditions, significant 𝜋-stacking tends to occur only at a later stage of film formation. At that point, however, the polymer chains have already become more entangled, and the adjacent 𝜋-stacked aggregates are more likely to interpenetrate and entangle with one another. This suppresses the formation of extended-chain 𝜋-stacks and reduces the degree of structural order in the resulting film. Consequently, films processed from a good solvent typically contain a larger fraction of disordered or amorphous regions.
These observations demonstrate that the solvent plays a decisive role in regulating polymer aggregation behavior. It influences not only the conformation and dispersion state of the polymer chains in solution, but also the onset, extent, and degree of order of aggregation during film formation. Therefore, solvent selection is a key factor in controlling polymer aggregation, and it can subsequently affect thin-film morphology and charge-transport properties.
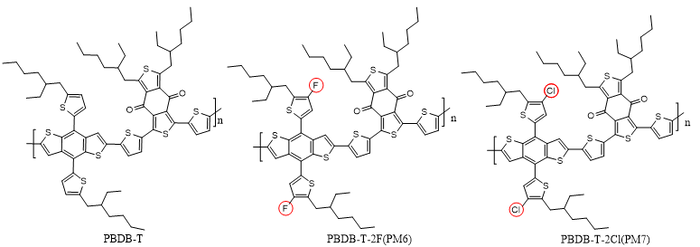
1.4 Material Choice[edit | edit source]
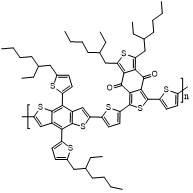
Solvent processing and substitution are two key factors governing the aggregation behavior of polymers, and are therefore closely related to their charge-transport properties. Accordingly, in order to systematically investigate the influence of aggregation on mobility, it is necessary to select a representative polymer system that can both reflect substitution effects and exhibit high sensitivity to processing conditions. On this basis, the PBDB-T family polymers were chosen as the model materials in this study, including PBDB-T, PM6 (PBDB-T-2F), and PM7 (PBDB-T-2Cl).
PM6 and PM7 are obtained from PBDB-T through halogen substitution, in which the two hydrogen atoms are substituted with fluorine and chlorine, respectively. This group of polymers, which share closely related molecular structures but differ in their substituents, provides an ideal model system for investigating the effects of halogen substitution on polymer aggregation and charge transport. In addition, these polymers are highly sensitive to solution-processing conditions. Their aggregation behavior, film-formation process, and resulting thin-film morphology can be readily modulated by solvent selection and processing treatment, making them particularly suitable for studying the relationship between solvent processing and aggregation. Finally, the PBDB-T family polymers are among the most representative donor polymers in the field of organic solar cells, and thus possess broad application relevance and substantial research value. Therefore, the use of the PBDB-T family as a model system not only helps elucidate the intrinsic relationship among substitution, solvent processing, aggregation, and mobility, but also ensures that the conclusions obtained are of greater general relevance and practical significance.
2. Project Aim[edit | edit source]
The aim of this study is to investigate how solvent type influence the aggregation behavior and charge-transport properties of PBDB-T family polymers, including PBDB-T, PM6, and PM7. In this work, the processing conditions include solvent type, solution concentration and thermal treatment. Specifically, chlorobenzene (CB) and chloroform (CF) are used as the processing solvents. Room temperature refers to 23°C, while the heating treatment in this study is applied only to the polymer solutions, with the solution temperature set at 100°C for CB and 60°C for CF. This work investigates how these factors effect solution-state aggregation, and further, by fabricating thin films from solutions with different aggregation states, to explore how these factors influence charge transport. Meanwhile, both the solution processing and film fabrication were carried out inside a glovebox to avoid interference from air exposure. To achieve this, hole-only diodes (HODs) based on these polymers are fabricated and analyzed through their current density-voltage (J-V) characteristics. Through this study, we expect to dig deeper regarding how molecular packing, aggregation, and film morphology collectively affect carrier mobility in conjugated polymer systems.
3. Experiment methods[edit | edit source]
3.1 Measurement of aggregation in different solvent systems[edit | edit source]
3.1.1 UV-Vis analysis of solution-state aggregation[edit | edit source]
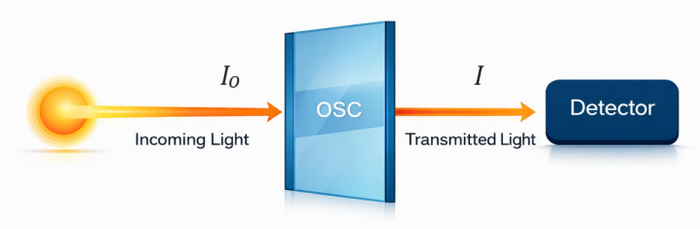
UV-Vis absorption spectroscopy is a simple and effective technique for testing aggregation behavior in polymer solutions[41][42]. The principle of UV-Vis is Beer-Lambert Law[43]: by comparing the incident light intensity I0 with the transmitted light intensity I, the transmittance T of the sample can be obtained as
The absorbance A is then given by
According to the Beer-Lambert law, the absorbance of a solution is proportional to the concentration 𝑐, the optical path length 𝑙, and the molar extinction coefficient
Here, is the molar extinction coefficient, 𝑙 is the optical path length, and 𝑐 is the solution concentration, 10mg/mL is the most commonly used concentration for film fabrication. Therefore, when the concentration and path length are kept as constant, changes in the UV-Vis spectrum mainly caused by solution aggregation.
For conjugated polymers, aggregation changes the intermolecular interactions and electronic coupling between polymer chains. These changes can be seen in the UV-Vis spectrums, such as shifts in peak position, changes in peak shape, and differences in the relative intensity of characteristic absorption features. Therefore, by comparing UV-Vis spectrums under different solvents, temperatures, the degree of aggregation in solution and its changes can be evaluated.
The solution-state aggregation of conjugated polymers is governed by multiple factors, including molecular structure, molecular weight, solvent, temperature, concentration, and additives. Among these, solvent and temperature are two of the most commonly varied processing parameters and have the most direct influence on chain conformation and interchain interactions in solution. Solvent affects the balance between polymer-solvent and polymer-polymer interactions, thereby determining whether the polymer chains remain molecularly dispersed or form pre-aggregates. Temperature effects the extent and ordering of aggregation by changing chain mobility and aggregate stability. Therefore, in this work, solvent and temperature are selected as the key variables to investigate the solution-state aggregation behavior of the PBDB-T family polymers, before examining how such aggregation influences film formation and carrier transport.
3.1.2 Variable-solvent UV–Vis measurements in solution: The sandwiched method[edit | edit source]
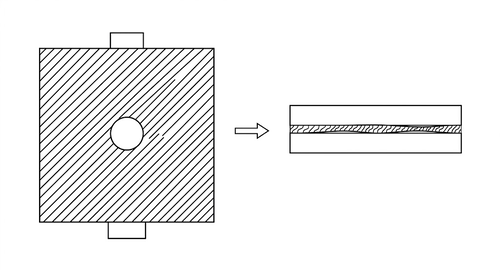
As shown in Fig 3.2, fixed temperature UV-Vis measurements were carried out on solutions prepared in different solvents in order to investigate the effect of solvent on solution-state aggregation, the setup shown in the figure. Two identical silicon substrates are selected and treated with oxygen plasma in order to improve surface wettability and ensure uniform spreading of the solution on the substrate surface. A piece of tape with a pre-cut hole in the center is taped tightly to one of the silicon substrates, after which an equal volume (40μL) of the test solution is deposited into the hole. The second silicon substrate is then placed on top as a cover. The two substrates are then tightly clamped along the edges with clips, which makes sure that solution has spread uniformly in the hole, and the optical path length of this part of solution is well-defined by the thickness of the tape and is consistent as per sample. After the sample is prepared, the solution UV-Vis was measured based on the principle shown in Fig. 3.1.
3.2 J-V curve measurement[edit | edit source]
3.2.1 Mott-Gurney Law: The method to calculate carrier mobility[edit | edit source]
In the SCLC regime, the current is mainly governed by bulk charge transport and follows the relation J∝V2. For a nearly trap-free unipolar device with efficient carrier injection, the current density can be described by the Mott–Gurney law:
where is the vacuum permittivity, is the relative permittivity, is the carrier mobility, Vbi is the built-in potential, and d is the thickness of the organic layer. Therefore, by fitting the J-V curve in the SCLC region using the Mott–Gurney law, the carrier mobility can be estimated.
The applicability of the Mott-Gurney law requires unipolar transport, which means only one type of charge carrier dominates during transport; ohmic injection; and no electrical traps within the selected voltage range. By blocking electron injection and ensuring that the device current is dominated by holes, the HOD structure avoids the complication of electron–hole recombination. As a result, the measured current more accurately reflects hole transport in the organic layer, making the extraction of carrier mobility from the J-V characteristics more reliable.
3.2.2 Selection of device type[edit | edit source]
Through UV–Vis experiments, we identified two factors that can affect solution aggregation: the solvent and halogen substitution. According to the study by Zhao et al., for annealed solutions, if spin-coating is carried out within a short period of time, the aggregation state in solution can be retained in the film state. As a result, the aggregation in these films differs from that of films prepared from solutions at room temperature. To further investigate the study’s objective, organic diodes were fabricated using solutions with different solvents and different annealing temperatures, and their J–V curves were measured.
To enable the application of the Mott–Gurney law for quantifying carrier mobility, hole-only diodes (HODs) were fabricated. In this device structure, electron injection is effectively blocked, so the device current is dominated by holes. Such a structure obeys the using conditions of the Mott–Gurney law, in which the current raise from single-carrier, space-charge-limited transport in the bulk of the organic layer, instead of from bipolar conduction or injection-limited processes at the interfaces. As a result, hole transport becomes the dominant process, allowing the hole mobility to be extracted more reliably. Therefore, this structure provides a more straightforward and reliable way to compare the effects of different materials and solvents on charge transport.
3.2.3 Selection of hole injection layer and cathode material[edit | edit source]
For the fabrication of hole-only diodes, the selection of the hole injection layer and cathode material is especially important. These two components determine the hole injection barrier and electron injection barrier, respectively, and therefore determine whether holes can be efficiently injected into the HOMO while electron injection is blocked by the electron injection barrier.
A HIL can facilitate hole injection into the HOMO of the organic layer by tuning the energy-level alignment at the electrode/organic interface and increasing the effective work function of the anode. For the hole injection layer, para-CF₃-substituted poly(triarylamine-alt-fluorene)(pTFF) is used in this study. According to the research of Png R.-Q. et al., heavily p-doped pTFF exhibits an ultrahigh vacuum work function as 5.9eV. As a result, the deep work function can significantly reduce the hole injection barrier, and it can provide efficient hole injection into the organic semiconductor. Therefore, pTFF is highly suitable as the HIL in this study.
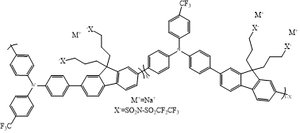
Among the metal materials commonly used as evaporated cathodes, Ag has a relatively deep work function, which helps increase the electron injection barrier and thereby effectively blocks electron injection. Therefore, Ag was chosen as the cathode in this study.
3.2.4 Processing of making organic diode[edit | edit source]
In this study, the p-doping process of pTFF was as follows. First, pTFF was weighed and heated at 120°C for at least 1 h to fully remove residual moisture. It was then dissolved in acetonitrile (ACN) to prepare a 15 mg/mL pTFF solution. Separately, an appropriate amount of Magic Blue was weighed and dissolved in ACN to prepare a 12 mM dopant solution. The dopant solution was then mixed with the pTFF solution at a doping level of at least 0.8 eq. After that, dimethyl carbonate(DMC) with a volume of at least three times that of the mixed solution was added. he mixture was centrifuged, and the supernatant was removed. This washing process was repeated twice to remove Na+ and SbCl6−. Then, an appropriate amount of ACN was added to redisperse the sample to a concentration of 20 mM. DMC with a volume of at least three times that of the solution was again added, followed by centrifugation, precipitation, and removal of the supernatant. This process was also repeated twice. Finally, an appropriate amount of ACN was added to prepare a 15 mg/mL solution, completing the doping process. The doped solution was processed into a film of about 30 nm, and the doping level was determined by UV–Vis spectroscopy. The doping level should be above 0.8. A 30nm film was fabricated from doped solution, the spin-coating condition is 2000rpm, 40s. The doping level was measured by UV–Vis spectroscopy.
During device fabrication, a p-doped pTFF solution was first prepared, and the spin-coating conditions were tested to obtain a uniform thin film. The pTFF film was then spin-coated, and its doping level and film thickness were measured. Before spin-coating film, the ITO substrates were pretreated as follows: The substrates were first marked and then cleaned with acetone and IPA, followed by oxygen plasma treatment for 10 mins to further remove residual contaminants and improve the surface condition. After these steps, the doped pTFF film was spin-coated onto the ITO substrates, and the film thickness was about 30 nm.
During the fabrication of the organic semiconductor active layer, OSC films were spin-coated onto ITO/pTFF substrates under different conditions. In total, 11 groups of devices were fabricated, as summarized in the table below. The thickness of the OSC films was approximately 120 nm. Finally, Ag was deposited as the cathode by thermal evaporation to complete the device fabrication.
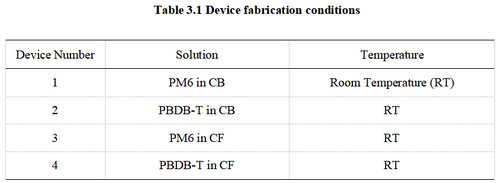

3.3 Electro Absorption Spectroscopy[edit | edit source]
3.3.1 Theory[edit | edit source]
Electro absorption technology is achieved by measuring the change in absorption coefficient after an applied electric field is applied[52][53][54][55]. Under normal incident conditions, the intensity I of light transmitted through the absorbing medium can be calculated using the Lambert-Beer law:
Where represents the original light intensity of the incident light, is the reflectivity, is the absorption coefficient, and is the thickness of the absorbing medium.
Both and are affected by the external electric field, and their changes as follows:
Under classical operating conditions, the change in the value of is negligible. Equation (3.6) simplifies to:
From a microscopic perspective, the energy level E(F) of state in electric field F is given by the following equation:
represents the electric dipole moment of this state, and is its polarizability. Therefore, the optical transition energy shift from the initial state to the final state is given by the following equation:
The first term represents the linear energy change caused by the difference in dipole moments between the initial and final states, which cancels out in isotropic solids without permanent dipoles. The second term describes the energy shift caused by the difference in polarizability between the initial and final states, an effect that always exists.
The absorption variation of the external electric field can be represented by the term of the Maclaurin series, while the third and higher order terms can be ignored due to the small spectral changes.
The sole contribution of the first term comes from the second-order Strac effect , while the isotropic average of in the second term does not cancel out, but instead produces in the randomly distributed matrix. The third contribution of comes from the transition of the oscillator to the previously forbidden state when the electric field is present, and this transition also has a quadratic relationship with F.
Adding up all contributions to , we get:
For a composite electric field with AC component and DC component :
Substituting formula (3.12) into (3.11) yields the modulation of at the fundamental frequencies and :
where and are the work functions of the two electrodes, q is the elementary charge, and d is the film thickness.
Using phase-sensitive lock-in detection, the harmonic components can be separated. Since ∆α(1ω) varies linearly with , the built-in field can be obtained by identifying the DC bias at which the 1ω electro absorption signal disappears. In this way, electro absorption spectroscopy provides a direct method for determining the built-in potential of the diode.
3.3.2 EA device setup and operation procedures[edit | edit source]
Before the EA measurement, the diode device was first connected to the cryostat inside the glovebox. The cryostat was then fixed on the holder, and the connection of the device was verified by measuring the J–V curve. After that, the cryostat was evacuated to 10−6mbar. Once the required pressure was reached, a pre-measurement was carried out to further confirm that the connection was normal, after which the system was cooled down. When the temperature inside the chamber reached 30 K, the formal EA measurement was performed.
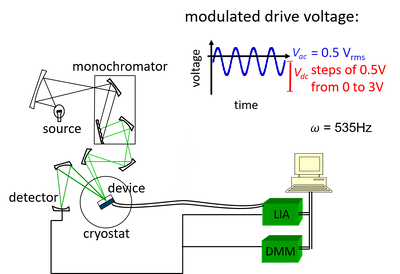

As shown in the schematic of the setup, direct measurement of the transmission signal is difficult due to the device structure. Therefore, in this experiment, the transmission signal was inferred from the measured reflection signal ∆R. This device is driven by a modulated driving voltage. The DC bias voltage varies from 0V to 3V in 0.5V steps to change the electric field strength, while a constant AC bias voltage is applied simultaneously. The change in electric field strength after applying this driving voltage leads to a change in the absorption bandgap ΔE.
Monochromatic light is incident on a glass substrate at a 45° angle, and the reflected light illuminates a photodiode. The voltage output of the photodiode is demodulated by a lock-in amplifier. This voltage output reflects the change in reflectivity with the excitation bias voltage Vdc within a certain photon energy range.
3.3.3 Method for calculating carrier mobility[edit | edit source]
By comparing the Vbi values of the CB solution system at RT and 60°C, it can be inferred that the Vbi at 100°C is close to the theoretical value of 1.2 V. Therefore, EA measurement for the 100°C devices is not necessary. After obtaining the specific Vbi of the device, the carrier mobility can be quantified using the Mott–Gurney law.
Although the Mott–Gurney law can directly provide an equation for carrier mobility, its application requires several ideal conditions, such as a uniform electric field, trap-free transport, and current dominated by SCLC.
Pasveer demonstrated that in disordered organic semiconductors, the carrier mobility μis dependent on both the local carrier density p and the electric field E. Neglecting these dependences may lead to an inaccurate estimation of carrier mobility. Therefore, a modified form of the Mott–Gurney law is required to obtain a more reliable value of carrier mobility. In this study, the derivation was carried out based on Gauss’s law and the drift–diffusion equation to better describe the physical processes in the device and obtain more reliable mobility values. The derivation proceeds as follows:
Gauss’s Law: , in 1D:
MG equation:
Drift-diffusion equation:
Integrate, we can get
Neglecting the term since we assume constant electrical field across OSC, then
From Gauss’s Law, we can calculate the carrier density:
The average carrier density is:
In Mott-Gurney regime:
4. Results and discussion[edit | edit source]
4.1 Results of UV-Vis[edit | edit source]
4.1.1 Result of variable-solvent[edit | edit source]
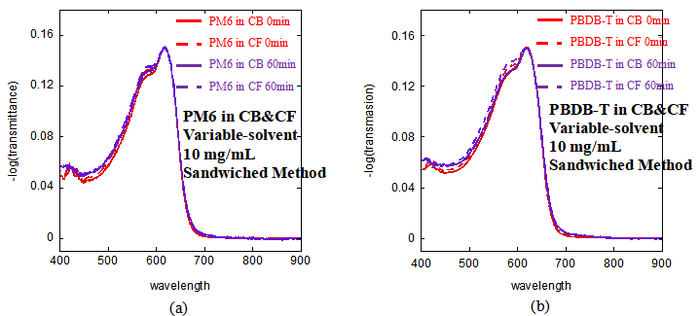
For both polymers, the difference between the two characteristic peaks is larger in CB than in CF. This suggests that the polymer chains show a stronger aggregation tendency in CB under the same conditions. The same trend is observed for both PM6 and PBDB-T, indicating that CB leads to a higher degree of solution-state aggregation than CF in these two polymer systems. In addition, no clear change is observed between the spectrums recorded at 0 min and 60 min for any of the four solutions. This indicates that all four solutions remain stable over the measured time range, and their aggregation states do not change significantly with time.
4.1.2 Conclusion for UV-Vis measurement[edit | edit source]
UV–Vis measurements were used to test the solution-state aggregation behavior of the PBDB-T family polymers and to evaluate the effects of solvent and temperature on their aggregation in solution. Under the same conditions, PM6 and PBDB-T show a stronger aggregation in CB than in CF. These results show that UV–Vis measurements can reveal the solution pre-aggregation state under different processing conditions before film deposition. Zhao[4] mentions that for annealed solutions, if spin-coating is carried out within a short period of time, the aggregation state in solution can be retained in the film state. So, the results further indicate that the initial aggregation state during film formation can be tuned by solvent selection and solution-heating conditions, thereby influencing the aggregation and microstructure of the resulting films.
4.2 Result of J-V curve[edit | edit source]
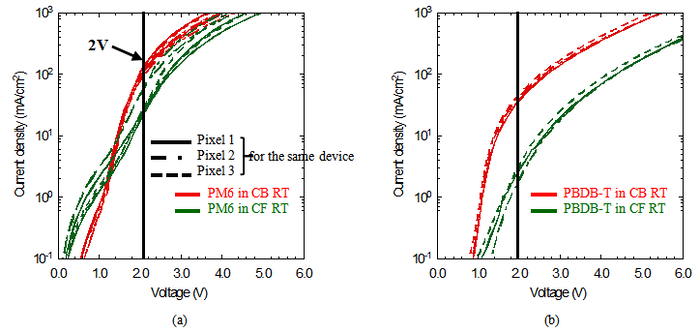
Since the forward and reverse scan curves are very similar, only the forward scan curves are shown in order to get clear results. According to the Mott–Gurney law, when the film thickness and applied voltage are the same, the carrier mobility μ is proportional to the current density J. Therefore, carrier mobility can be estimated by comparing the current density of films with the same thickness at the same voltage.
As shown in Fig. 4.2, at the same temperature, the mobility of PM6 films processed from CB is higher than that of films processed from CF, while the opposite trend is observed for PBDB-T. These results indicate that solvent processing conditions can significantly influence the carrier mobility of the films.
Because the above conclusion was reached under the assumption that different devices have the same film thickness and built-in potential, it may be inaccurate if the actual device thickness or built-in potential varies. Therefore, electro absorption(EA) spectroscopy is required to determine of each device, so that carrier mobility can be quantified using the Mott-Gurney law.
4.3 Result of EA[edit | edit source]
From the theory of EA, of different device can be measured. According to EA theory, the null bias is the DC bias that cancels the built-in electric field in the device. At this bias, the Stark shift becomes zero, so this voltage is taken as . In this study, as increases, the EA feature changes from a peak to a dip, which happens near 1.85eV. The null bias is taken as the voltage between these two cases, where the feature becomes almost flat. This voltage is defined as the built-in potential . Here is the result of EA:
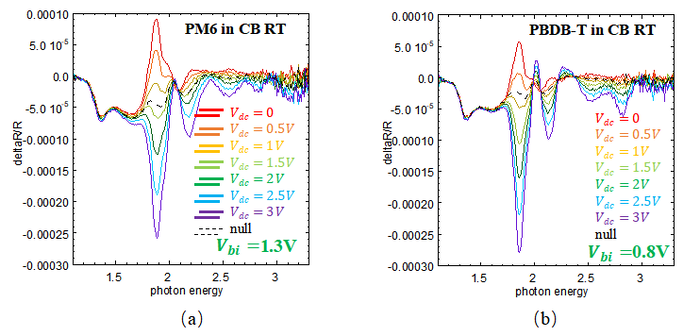
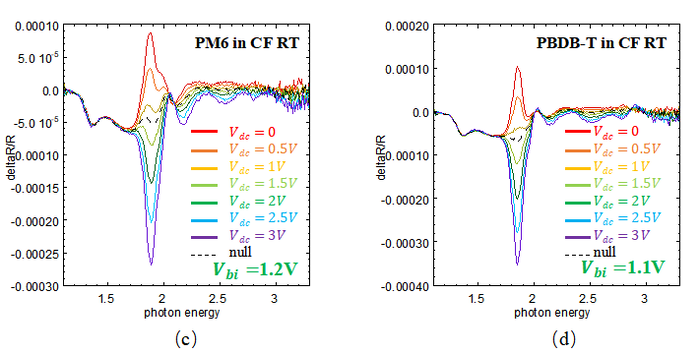
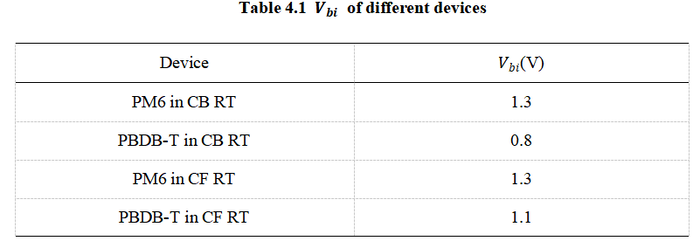
4.4 Result of carrier mobility[edit | edit source]
Based on the above equations, the carrier mobility was calculated using the J–V curve data, and the results are shown below.
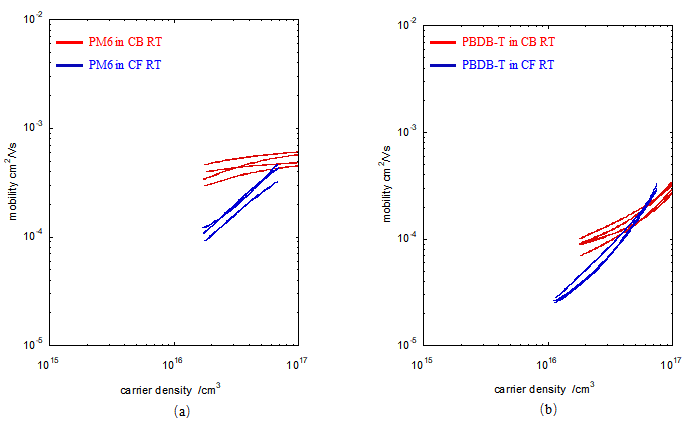
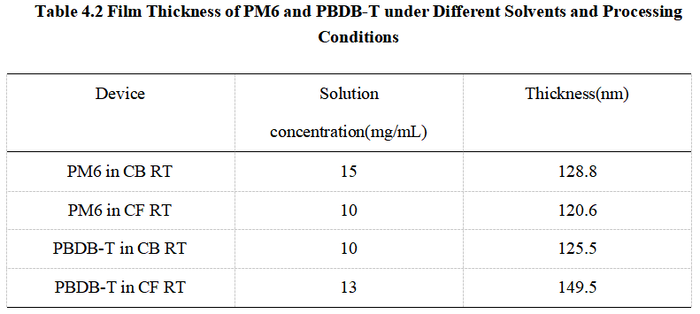
As shown in Figure 4.4, solvent type has a significant influence on charge carrier mobility. For films of the same polymer measured under similar conditions, the mobility of the CB-based film is much higher than that of the CF-based film. This result indicates that solvent processing can change the solution-state aggregation and thereby affect charge transport. For both PBDB-T and PM6, CB is a more suitable solvent than CF. In the CB system, both polymers show better solubility and stronger aggregation in solution, and the corresponding films exhibit higher carrier mobility. These results suggest that, within the processing range studied here, solvent-dependent aggregation plays a key role in determining charge-transport behavior.
Conclusion[edit | edit source]
This study systematically investigates how solvent type affects the aggregation behavior and charge transport of PBDB-T family polymers, and further explores how solvent-controlled aggregation influences molecular packing and film morphology. The J–V curves and the extracted carrier mobility show that solvent type strongly affects charge transport. The experimental results demonstrate that, among the solvent-processing parameters studied here, solvent type plays the dominant role in determining carrier mobility. In particular, films prepared from CB exhibit much higher carrier mobility than those prepared from CF. By relating solution-state aggregation to carrier mobility, the results further suggest that solvent-dependent aggregation plays a critical role in charge transport.
A possible explanation is that CB promotes a more favorable aggregation state in solution, which provides a certain degree of molecular pre-organization before or during film formation. This pre-organization helps establish more ordered π–π stacking, enhances intermolecular electron cloud overlap, and enables more continuous charge-transport pathways, thereby leading to higher carrier mobility. In contrast, CF leads to a less favorable aggregation state, which is less effective in supporting ordered molecular packing and continuous transport pathways.
Overall, this study shows that charge transport in organic semiconductors can be strongly influenced by solvent type. The effect of solvent type is not limited to the solution state, but extends to molecular packing, film morphology, and the continuity of charge-transport pathways in the solid film. Therefore, controlling aggregation through solvent selection provides an effective strategy for improving charge transport in PBDB-T family polymer systems.
References[edit | edit source]
[1].Koh, Q. M., Mazlan, N. S., Seah, Q. J., Yang, J. C., Chen, Y. J., Png, R. Q., Ho, P. K. H. & Chua, L. L., ACS Appl Mater Interfaces 2024, 16, 39708-39716.
[2].Sachnik, O., Tan, X., Dou, D., Haese, C., Kinaret, N., Lin, K. H., Andrienko, D., Baumgarten, M., Graf, R., Wetzelaer, G. A. H., Michels, J. J. & Blom, P. W. M., Nat Mater 2023, 22, 1114-1120.
[3].Ang, Adele A.H., Xiong, X.C., Yang, J.C., Hong, Z.L., Png, R.Q., Chua, L.L., Ho, Peter K.H. (unpublished).
[4].Zhao, L. H., Ping, R. Q., Zhuo, J. M., Wong, L. Y., Tang, J. C., Su, Y. S. & Chua, L. L., Macromolecules 2011, 44, 9692–9702.
[5].Li, Y., Yan, S., Geng, M. & Li, H., Enhancing π–π stacking by a halogen substituent in a single-molecule junction, Chem. Commun. 2025, 61, 8419–8422.
[6].Sirringhaus, H., Tessler, N. & Friend, R. H., Integrated optoelectronic devices based on conjugated polymers, Science 1998, 280, 1741–1744.
[7].Facchetti, A., π-Conjugated polymers for organic electronics and photovoltaic cell applications, Chem. Mater. 2011, 23, 733–758.
[8].Forrest, S. R., The path to ubiquitous and low-cost organic electronic appliances on plastic, Nature 2004, 428, 911–918.
[9].Sirringhaus, H., Device physics of solution-processed organic field-effect transistors, Adv. Mater. 2005, 17, 2411–2425.
[10].Zhang, S., Qin, Y., Zhu, J. & Hou, J., Over 14% efficiency in polymer solar cells enabled by a chlorinated polymer donor, Adv. Mater. 2018, 30, 1800868.
[11].Hou, J., Inganäs, O., Friend, R. H. & Gao, F., Organic solar cells based on non-fullerene acceptors, Nat. Mater. 2018, 17, 119–128.
[12].Facchetti, A., π-Conjugated polymers for organic electronics and photovoltaic cell applications, Chem. Mater. 2011, 23, 733–758.
[13].Blom, P. W. M. & de Jong, M. J. M., Electrical characterization of organic semiconductor devices, IEEE J. Sel. Top. Quantum Electron. 2000, 6, 1059–1068.
[14].Brütting, W., Adachi, C., Physics of Organic Semiconductors, Wiley-VCH, 2012.
[15].Zhou, M., Energy Level Alignment in Semiconducting Organic Electronic Devices, Ph.D. Dissertation, National University of Singapore, Singapore, 2010.
[16].Blom, P. W. M. & de Jong, M. J. M., Electrical characterization of organic semiconductor devices, IEEE J. Sel. Top. Quantum Electron. 2000, 6, 1059–1068.
[17].Mott, N. F. & Gurney, R. W., Electronic Processes in Ionic Crystals, Oxford University Press, Oxford, 1940.
[18].Zhou, M., Chua, L.-L., Png, R.-Q., Yong, C.-K., Sivaramakrishnan, S., Chia, P.-J., Wee, A. T. S., Friend, R. H. & Ho, P. K. H., Hole-Doped Interfaces at Ohmic Contacts to Organic Semiconductors, Phys. Rev. Lett. 2009, 103, 036601.
[19].Haneef, H. F., Zeidell, A. M. & Jurchescu, O. D., Charge Carrier Traps in Organic Semiconductors: A Review on the Underlying Physics and Impact on Electronic Devices, J. Mater. Chem. C 2020, 8, 759–787.
[20].Liu, Z. & Lin, Y., Testing Trap States in Polymer Solar Cells, Polym. Test. 2024, 108387.
[21].Zhao, C., Tang, C. G., Seah, Z.-L., Koh, Q.-M., Chua, L.-L., Png, R.-Q. & Ho, P. K. H., Improving organic photovoltaic cells by forcing electrode work function well beyond onset of Ohmic transition, Nat. Commun. 2021, 12, 2358.
[22].Urbančič, J., Charge Transport in Organic Semiconductors, IntechOpen, 2025.
[23].Zhao, N., Osman, K., Png, R. Q., Ho, P. K. H. & Chua, L.-L., Effect of side-chain length on the structure and charge transport in conjugated polymers, J. Am. Chem. Soc. 2010, 132, 15362–15363.
[24].Aasmundtveit, K. E.; Samuelsen, E. J.; Guldstein, M.; Steinsland, C.; Flornes, O.; Fagermo, C.; Seeberg, T. M.; Pettersson, L. A. A.; Inganäs, O.; Feidenhans’l, R.; Ferrer, S. Macromolecules 2000, 33, 3120-3127.
[25].Chang, J. F.; Sun, B.; Breiby, D. W.; Nielsen, M. M.; Sölling, T. I.; Giles, M.; McCulloch, I.; Sirringhaus, H. Chem. Mater. 2004, 16, 4772-4776.
[26].Zhao, L.-H.; Png, R.-Q.; Zhuo, J.-M.; Wong, L.-Y.; Tang, J.-C.; Su, Y.-S.; Chua, L.-L. Role of Borderline Solvents to Induce Pronounced Extended-Chain Lamellar Order in π-Stackable Polymers. Macromolecules 2011, 44 (24), 9692–9702.
[27].Chang, J. F.; Clark, J.; Zhao, N.; Sirringhaus, H.; Breiby, D. W.; Andreasen, J. W.; Nielsen, M. M.; Giles, M.; Heeney, M.; McCulloch, I. Phys. Rev. B 2006, 74, 115318-1-12.
[28].Samitsu, S.; Shimomura, T.; Heike, S.; Hashizume, T.; Ito, K. Macromolecules 2008, 41, 8000-8010.
[29].Kim, D. H.; Park, Y. D.; Jang, Y.; Kim, S.; Cho, K. Macromol. Rapid Commun. 2005, 26, 834-839.
[30].Merlo, J. A.; Frisbie, C. D. J. Polym. Sci., B: Polym. Phys. 2003, 41, 2674-2680.
[31].Wang, S.; Tang, J. C.; Zhao, L. H.; Png, R. Q.; Wong, L. Y.; Chia, P. J.; Chan, H. S. O.; Ho, P. K. H.; Chua, L. L. Appl. Phys. Lett. 2008, 93, 162103-1-3.
[32].Sperling, L. H. Introduction to Physical Polymer Science, 3rd ed.; Wiley-Interscience: New York, 2001.
[33].Tan, Z.-S., Jamal, Z., Teo, D. W. Y., Ko, H.-C., Seah, Z.-L., Phua, H.-Y., Ho, P. K. H., Png, R.-Q. & Chua, L.-L., Optimization of fluorinated phenyl azides as universal photocrosslinkers for semiconducting polymers, Nat. Commun. 2024, 15, 6354.
[34].Png, R.-Q., Chia, P.-J., Tang, J.-C., Liu, B., Sivaramakrishnan, S., Zhou, M., Khong, S.-H., Chan, H. S. O., Burroughes, J. H., Chua, L.-L., Friend, R. H. & Ho, P. K. H., High-performance polymer semiconducting heterostructure devices by nitrene-mediated photocrosslinking of alkyl side chains, Nat. Mater. 2010, 9, 152–158.
[35].Zhang, Q., Kelly, M. A., Bauer, N. & You, W., The Curious Case of Fluorination of Conjugated Polymers for Solar Cells, Acc. Chem. Res. 2017, 50, 2401–2409.
[36].Wheeler, S. E., Understanding Substituent Effects in Noncovalent Interactions Involving Aromatic Rings, Acc. Chem. Res. 2013, 46, 1029–1038.
[37].Kim, I.-B., Jang, S.-Y., Kim, Y.-A., Kang, R., Kim, I.-S., Ko, D.-K. & Kim, D.-Y., The Effect of Fluorine Substitution on the Molecular Interactions and Performance in Polymer Solar Cells, ACS Appl. Mater. Interfaces 2017, 9, 24011–24019.
[38].Leclerc, N., Chávez, P., Ibraikulov, O. A., Heiser, T. & Lévêque, P., Impact of Backbone Fluorination on π-Conjugated Polymers in Organic Photovoltaic Devices: A Review, Polymers 2016, 8, 11.
[39].Zheng, Z., Yao, H., Ye, L., Xu, Y., Zhang, S. & Hou, J., PBDB-T and its Derivatives: A Family of Polymer Donors Enables over 17% Efficiency in Organic Photovoltaics, Mater. Today 2020, 35, 115–130.
[40].Grüne, J., Stähly, B., Lulei, S., Kotova, M., Dyakonov, V., Gillett, A. J. & Sperlich, A., Triplet Excitons and Associated Efficiency-Limiting Pathways in Organic Solar Cell Blends Based on Non-Halogenated PBDB-T and Y-Series, Fluorinated PM6:Y6, and Chlorinated PM7:Y7, Adv. Funct. Mater. 2023, 33, 2212640.