Alcohol Concentration Measurement: Difference between revisions
No edit summary |
No edit summary |
||
| Line 10: | Line 10: | ||
This research proposes a non-contact alcohol concentration measurement method and infrared spectroscopy. This method measures changes in infrared light intensity transmitted through alcohol solutions, using photoelectric sensors and data fitting techniques to achieve high precision measurements. | This research proposes a non-contact alcohol concentration measurement method and infrared spectroscopy. This method measures changes in infrared light intensity transmitted through alcohol solutions, using photoelectric sensors and data fitting techniques to achieve high precision measurements. | ||
[[File:Transmittance-Diagram-1024x447.png|center|thumb|Schematic Diagram of the Experiment]] | [[File:Transmittance-Diagram-1024x447.png|center|thumb|Schematic Diagram of the Experiment]] | ||
== Advantages of Infrared-Based Alcohol Detection == | |||
* '''Non-contact measurement''', reducing contamination. | |||
* '''High accuracy''', based on alcohol-specific infrared absorption characteristics. | |||
* '''Low cost''', with simple photodetection devices. | |||
== Equipment Required == | == Equipment Required == | ||
Revision as of 10:53, 1 April 2025
Experiment to Measure Alcohol Concentration Using Oband Light Source and a Photodetector
Team Member
Lim Gin Joe, Sun Weijia, Yan Chengrui, Zhu Junyi
Background
Alcohol (ethanol) is a widely used organic compound in medicine, chemical industry, food, and beverages. Rapid and accurate measurement of alcohol concentration is crucial in various industries, including food and beverage manufacturing, medical disinfection, and industrial synthesis. Existing measurement methods typically have limitations such as complexity, high equipment costs, and large measurement errors. Therefore, developing an efficient, cost-effective, and non-contact measurement method is essential.
Objective
This research proposes a non-contact alcohol concentration measurement method and infrared spectroscopy. This method measures changes in infrared light intensity transmitted through alcohol solutions, using photoelectric sensors and data fitting techniques to achieve high precision measurements.
Advantages of Infrared-Based Alcohol Detection
- Non-contact measurement, reducing contamination.
- High accuracy, based on alcohol-specific infrared absorption characteristics.
- Low cost, with simple photodetection devices.
Equipment Required
- Oband Light Source: A broadband light source capable of emitting light across a spectrum of wavelengths.
- Photodetector: A device to measure the intensity of light that has passed through the sample solution.
- Alcohol Samples: Different alcohol solutions with known concentrations (e.g., ethanol, isopropanol).
- Cuvette: A transparent container to hold the alcohol solution for light transmission.
- Spectrometer: For recording and analyzing the intensity of transmitted light.
- Power Supply: To power the Oband light source.
Methodology
1. Setup the Apparatus
- Position the Oband Light Source: Place the Oband light source so that the emitted light can be directed towards the alcohol sample in the cuvette.
- Align the Photodetector: Position the photodetector on the opposite side of the cuvette to measure the intensity of light that passes through the alcohol solution.
- Cuvette Setup: Fill the cuvette with the alcohol sample. Ensure that the path of light through the cuvette is clear and that the cuvette is free from air bubbles.
- Ensure Proper Calibration: Before measuring the alcohol samples, calibrate the setup by using a solution of known concentration or distilled water to establish a baseline (zero absorption).
2. Measure Absorbance
- Test Different Concentrations: Prepare alcohol samples of known concentrations. This will allow you to measure the light intensity passing through different levels of alcohol and correlate the intensity with the concentration.
- Record Light Intensity: For each sample, measure the light intensity that reaches the photodetector. Record the values using the spectrometer. Ensure to measure the intensity multiple times for each concentration to ensure accuracy.
3. Analyze the Results
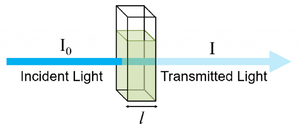
- Calculate Absorbance: Use the formula:
where is absorbance, is the intensity of light from the Oband source, and is the intensity of light received by the photodetector after passing through the alcohol solution.
- Plot the Absorbance vs Concentration: Create a graph to visualize the relationship between absorbance and alcohol concentration. The graph should show a linear or near-linear correlation.
- Determine the Alcohol Concentration: Using the absorption curve and the known concentration values, estimate the alcohol concentration of an unknown sample by comparing its absorbance value.
Data Analysis
1. Beer-Lambert Law: The relationship between absorbance and concentration is governed by the Beer-Lambert Law, which states:
Where: * is the absorbance, * is the molar absorptivity (a constant for the alcohol), * is the concentration of the alcohol, * is the path length of light through the sample.
References
- Beer-Lambert Law for Spectrophotometry
- Applications of Optical Sensing for Chemical Analysis
- Practical Guide to Spectrometer Calibration